Found 1 results
Article
14 November 2024The Discovering of Rapid Formation La(OH)3 from LaH3
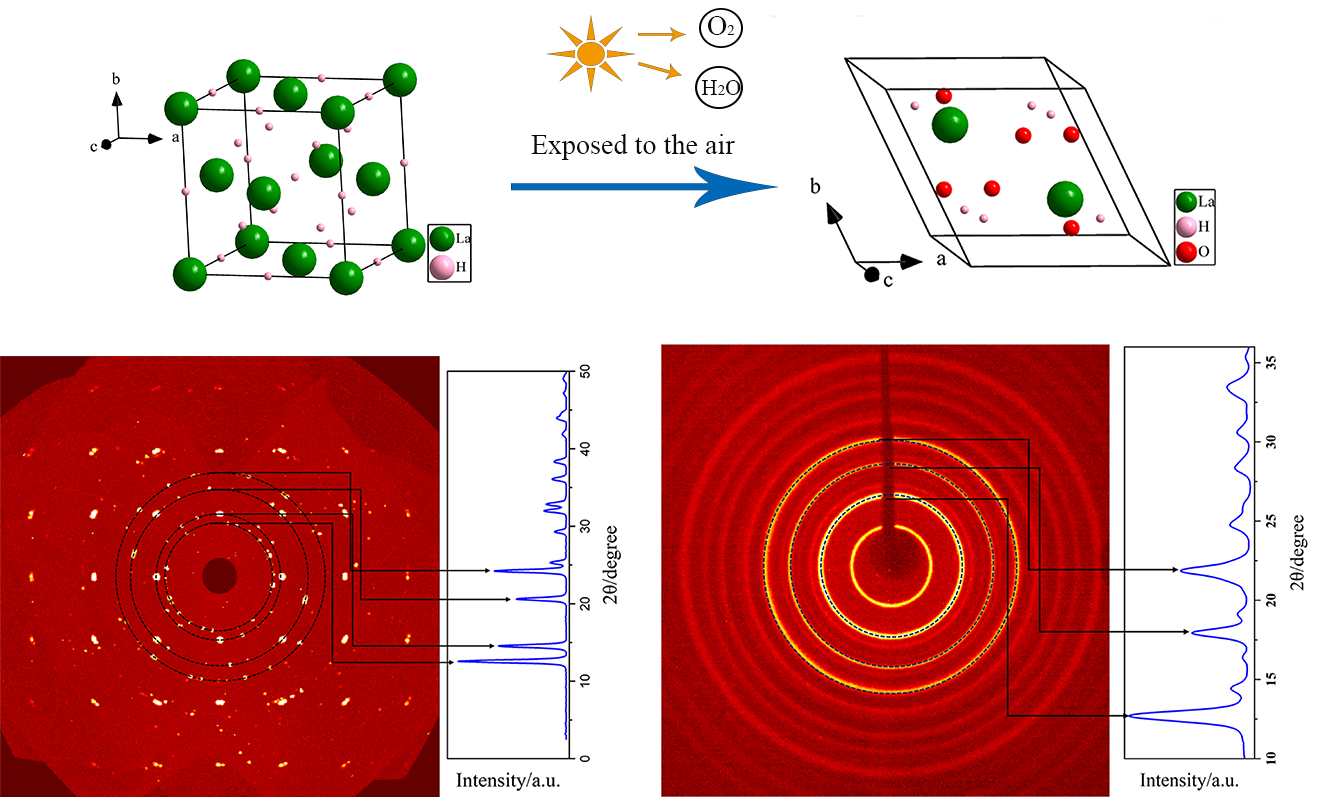
It was found that the single crystal of LaH3 specimen with $${Fm\overline{3}m}$$ (No.225) will decompose into powders within 24 h, which is later characterized to be La(OH)3 by single crystal X-ray diffraction (SXRD) measurements. The discovery motivates the examination of three possible transition paths by comparing formation enthalpy with first-principles calculations and employing a custom- designed hydrogen detection setup. Furthermore, the most suitable adsorption position of O2 molecules on the (111) surfaces has been investigated by comparing the adsorption enthalpy from different candidate positions by utilizing first-principles calculations, implying the pivotal role of O2 molecules played in the rapid formation of La(OH)3 along the optimal transition path.