Contents
Evaluation of the Efficacy of Chinese Inactivated COVID-19 Vaccines against the Delta Variant in the Nanjing Outbreak: A Cohort Study
Fuqun Liu
1,†
Shufei Wu
2,3,†
Mengying Liu
2
Lili Wang
1
Xinmei Huang
2
Fuchao Li
4
Weihua Wu
4
Ying Xu
4
Zhigang Zhao
4
Yangyang Xia
4
Yujuan Wang
4
Yue Yang
4
Wei Wang
1
Haisen Zhou
1,*
Mengshu Cao
2,3,*
Author Information
Other Information
1
Department of Rheumatology and Immunology, Lishui District Traditional Hospital of Chinese Medicine, Nanjing University of Chinese Medicine, Nanjing 211200, China
2
Department of Respiratory and Critical Care Medicine, Nanjing Drum Tower Hospital, The Affiliated Hospital of Nanjing University Medical School, Nanjing 210008, China
3
Department of Respiratory and Critical Care Medicine, Nanjing Drum Tower Clinical Medical College, Nanjing University of Chinese Medicine, Nanjing 210008, China
4
Nanjing Drum Tower Hospital, The Affiliated Hospital of Nanjing University Medical School, Nanjing 210008, China
*
Authors to whom correspondence should be addressed.
†
These authors contributed equally to this work.
Received: 20 August 2024 Accepted: 20 September 2024 Published: 27 September 2024
© 2024 The authors. This is an open access article under the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/).
J. Respir. Biol. Transl. Med.
2024,
1(3), 10016;
DOI: 10.70322/jrbtm.2024.10016
ABSTRACT:
Background: The strains of COVID-19 are constantly mutating,
and the effectiveness of Chinese inactivated vaccines against the COVID-19
Delta variant has not been described clearly. Methods: The clinical data of
patients with the COVID-19 Delta variant in the 2021 Nanjing outbreak were
retrospectively reviewed. Results:
There were 212 patients with the COVID-19 Delta variant (unvaccinated, n = 56,
26.42%; vaccinated, n = 156, 73.58%) included in our cohort study. The median
age was 45.5 (38, 53) years old. Eighty-seven subjects (41.04%) were airport
staff, and 94 patients (44.34%) in 32 families were infected. There were 53
(25.00%) and 103 (48.58%) cases with one-dose and two-dose vaccination,
respectively, and 55 (25.94%), 147 (69.34%) and 10 (4.72%) had mild, moderate
and severe symptoms, respectively. The duration of viral shedding, or viral
shedding time (VST), was significantly longer in unvaccinated individuals compared
to vaccinated individuals (p = 0.0008). Moreover, the duration was significantly
longer in patients who received one vaccine dose than those who received two
doses (p < 0.0001).
The mild patients had significantly shorter VSTs than the moderate subjects (p < 0.0001).
Disease severity and vaccination dose were independent predictors for VST by
Cox regression models. Conclusions: These
results suggest that two-dose vaccination could reduce VST in patients with the
COVID-19 Delta variant. Chinese inactivated vaccines may decrease the disease
severity of cases with the COVID-19 Delta variant.
Keywords:
COVID-19; Delta variant; Chinese inactivated COVID-19 vaccine
1. Introduction
The emergence of the coronavirus disease 2019 (COVID-19) has led to a global crisis with significant consequences. The latest reports from the World Health Organization (WHO), as of 13 August 2024, revealed a total of 541 million confirmed cases and nearly 6.3 million deaths [1]. COVID-19 is caused by SARS-CoV-2, an RNA virus frequently mutates during replication. Among the countless mutations that have occurred, several variants of concern (VOCs) have been specifically identified, namely: Alpha (B.1.1.7), Beta (B.1.351), Gamma (P.1), Delta (B.1.617.2) and Omicron (B.1.1.529) [2,3]. B.1.617.2 was initially reported in India and then spread worldwide in 2021. It possesses 2 key mutations (L452R and E484Q) in the receptor-binding domain, enabling it to evade neutralizing antibodies [4]. In comparison to the alpha and beta variants, the delta variant results in higher viral loads and higher transmissibility [5].
Vaccination plays a pivotal role in effectively safeguarding individuals against severe COVID-19 by stimulating the production of neutralizing antibodies and potentially inducing cytotoxic T lymphocytes [6]. However, it is crucial to acknowledge that the Delta variant exhibits a diminished sensitivity to serum-neutralizing antibodies derived from recovered individuals and those who have received COVID-19 vaccines compared to other variants at the time of this study [7]. Consequently, unvaccinated individuals face a higher risk of hospitalization and requiring emergency medical care [8]. Nevertheless, it is important to highlight that COVID-19 vaccines offer substantial protection against the Delta variant when individuals complete their full vaccination regimen. Studies indicate that achieving full vaccination can potentially reduce the risk of infection and symptomatic disease by approximately 20–30% after a period of 6 months [9,10]. Inactivated COVID-19 vaccines that were developed in China have consistently shown efficacy in combination with good safety and tolerability profiles. Similar encouraging results have also been reported in trials conducted in Turkey [11,12,13].
Despite the extensive research conducted on COVID-19 vaccines, there remains a significant gap in our understanding of the duration required for viral clearance in individuals who have received inactivated vaccines. To bridge this knowledge gap, our study aimed to fill this void by analyzing the clinical data of 212 patients infected with the Delta variant of COVID-19. Our primary objective was to assess the effectiveness of Chinese COVID-19 vaccines against the Delta variant by examining viral shedding time (VST) during the Nanjing outbreak in the summer of 2021. Through this research, we aim to contribute to a more comprehensive understanding of the efficacy of Chinese COVID-19 vaccines in combatting the Delta variant, thus informing future vaccination strategies and public health interventions.
2. Materials and Methods
2.1. Study Population
This cohort study included 212 patients with the COVID-19 Delta variant from Lishui District Traditional Hospital of Chinese Medicine in the summer of 2021. All patients were transferred from the Nanjing Public Health Center for centralized observation. They met the following discharge criteria from the Nanjing Public Health Center: ① Their temperature was normal for more than 3 days; ② Their respiratory symptoms and acute lung lesions on chest imaging were improved; and ③ Their tests for COVID-19 mRNA were negative two times at least every 24 h [14]. The participants in this study were divided into two main groups: the unvaccinated group and the vaccinated group. The vaccinated group was further stratified into two subgroups based on the number of vaccine doses received: one-dose vaccinated and two-dose vaccinated subgroups. All vaccines administered to the patients in this study were inactivated vaccines manufactured by Chinese companies, including Sinovac Life Sciences Co., Ltd., Beijing, China, Beijing Institute of Biological Products Co., Ltd., Beijing, China and Anhui Zhifei Longcom Biopharmaceutical Co., Ltd., Hefei, China. The study protocol was reviewed and approved by the Ethics Committee of Lishui District Traditional Hospital of Chinese Medicine in accordance with the principles outlined in the Declaration of Helsinki (1989) (Approval No. 2021LCGC001). Prior to their participation, all patients provided informed consent by signing the consent forms.
2.2. Clinical Patterns of Disease Severity
The confirmed positive patients were classified into four categories of disease severity (mild, moderate, severe, and critical) according to the updated eighth version of China's COVID-19 prevention and control protocol [14]. The mild clinical pattern was defined as mild clinical symptoms, and there was no pneumonia on chest imaging. The moderate clinical pattern presented as fever, respiratory system symptoms, and pneumonia on chest imaging. The severe pattern needed to meet any of the following criteria: respiratory rate >30/min, resting oxygen saturation <93%, and oxygenation index <300 mmHg. The critical pattern included any of the following conditions: respiratory failure and requiring mechanical ventilation, shock or other organ failure requiring admission to the ICU [14,15]. Regarding the above items, our study had no patients with critical patterns.
2.3. Definition of COVID-19 Delta Variant Shedding Time
The cycle threshold (CT) value of COVID-19 Delta mRNA lower than (<) 35 was identified as positive, and above (≥) 35 was considered negative by real-time polymerase chain reaction (RT‒PCR) tests with samples of oropharyngeal or nasopharyngeal swabs. All patients in Lishui Hospital of Traditional Chinese Medicine were transferred from Nanjing Public Health Center if they met the following three criteria [14]. They needed to take at least 3 tests for COVID-19 mRNA during the following 2 weeks (the 7th, 12th and 13th days after the transfer). They would have to be isolated for another 13 days with another 3 tests if one test came back positive. Until they had three negative tests on the 7th, 12th and 13th days after their last positive COVID-19 mRNA test, they were transferred to a hotel for another 2 weeks of observation with a COVID-19 mRNA test once a week. If these tests were negative, they would go home and continue observation for one month, taking tests once a week. If any test while in the hotel was found to be positive, they would transfer to the Nanjing Public Health Center again for additional isolation and tests according to the above rules.
The VST, which is the time from positive to negative mRNA tests, was defined from the first day of a positive test to the following 13th day or the last day of a positive test plus an additional 13 days (Figure 1A–D).
The flow chart of cases transferred and negative or relapsing-positive mRNA tests is shown in Supplemenatry Figure S1. There were 143 (67.45%) cases relapsed to positive and 9 (4.25%) without an exact VST due to loss of follow-up in our study.
2.4. Clinical Data Collection
The demographic data (sex, age, smoking history and occupational exposure), COVID-19 vaccination (date, doses of vaccination and manufacturer), epidemiology (family cluster outbreak and date of each COVID-19 mRNA test) and clinical characteristics (symptoms, clinical pattern of disease severity and complication) were collected from the electronic medical records (EMR) and follow-up by telephone. The deadline for follow-up was 31 December 2021. No cases had died from COVID-19 infection by the end of the follow-up. Two senior radiologists reviewed and evaluated the radiological findings of chest computed tomography (CT) separately.
2.5. Statistical Analysis
Continuous variables are presented as the median (interquartile range) or mean ± standard deviation (SD), and categorical variables are expressed as numbers (%). Categorical variables were assessed by chi-square (χ2) or Fisher exact tests, and continuous variables were evaluated by independent samples t tests, Mann‒Whitney U tests or one-way ANOVA tests. The bivariate correlation analysis between the number of vaccinations and disease severity was assessed using a Spearman correlation test. Univariate and multivariate Cox regression models were performed to examine the predictors of the time from positive to negative COVID-19 mRNA after diagnosis with a p-value < 0.05. Statistical analyses were performed using Prism version 9 (GraphPad, San Diego, CA, USA). p < 0.05 was considered statistically significant.
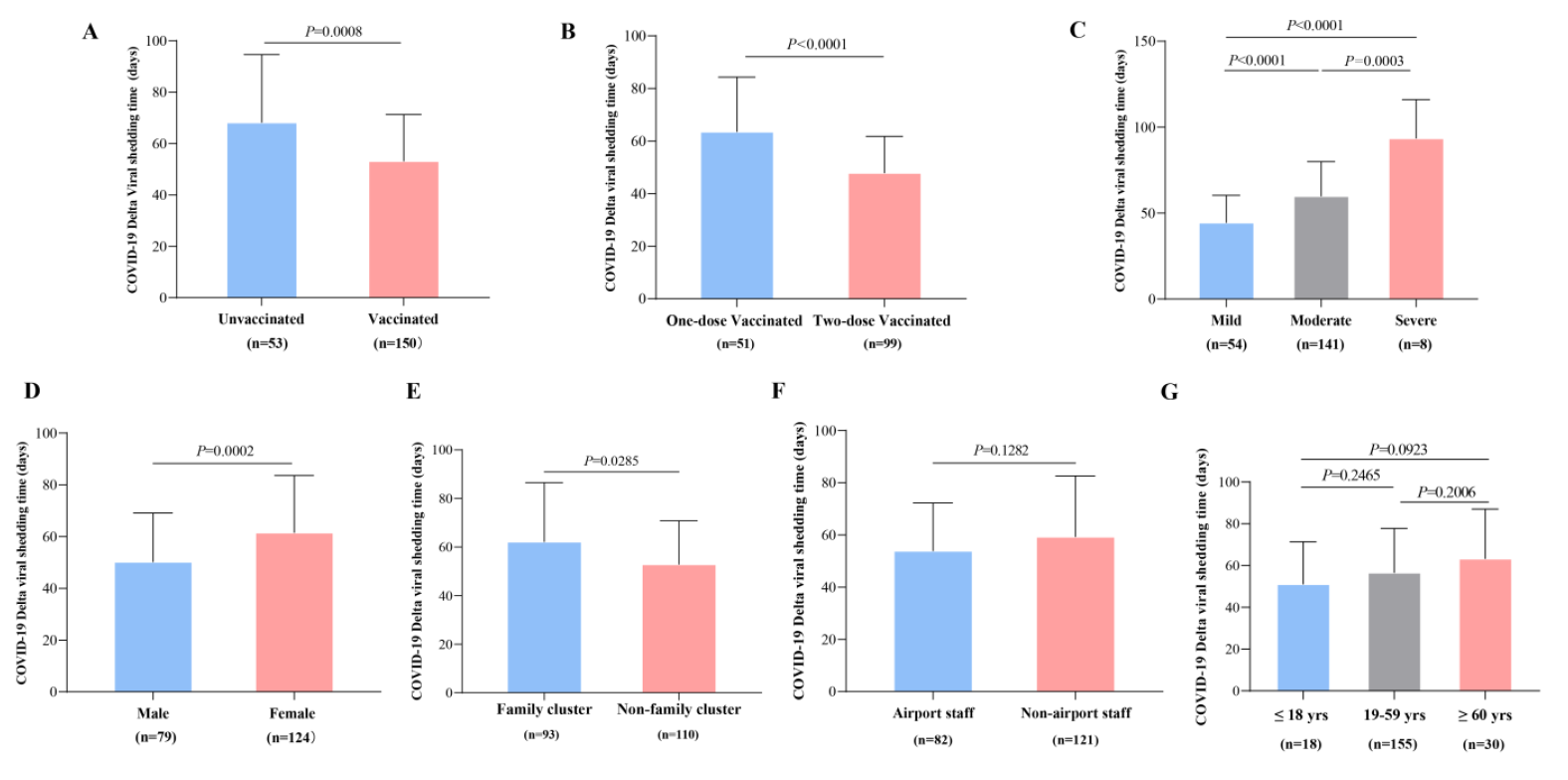
Figure 1. <b>Definition of COVID-19 Delta Viral Shedding Time (VST).</b> VSTs and transfer locations for (<b>A</b>) 69 individuals without re-positive results; and 143 individuals with re-positive results: (<b>B</b>) 87 in hospital, (<b>C</b>) 19 in hotel, and (<b>D</b>) 37 in both settings.
3. Results
3.1. Time-Series Plots of COVID-19 Patients with Delta Variants in the 2021 Summer Nanjing Outbreak
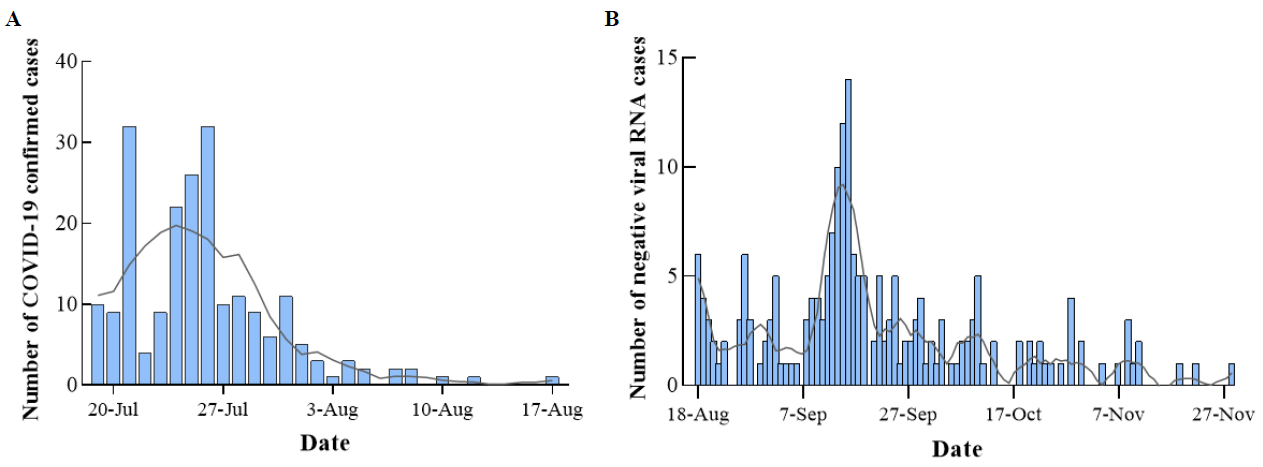
The Nanjing outbreak of COVID-19 began on 19 July 2022. The daily increase in the number of patients peaked on 26 July, and community transmission was stopped on 17 August. This local epidemic of patients with positive COVID-19 lasted for a month (Figure 2A). After 18 August, the COVID-19 mRNA tests of patients gradually became negative, and the negative cases peaked after 1 month. All cases were negative by 27 November (Figure 2B). The epidemic outbreak in the airport ended after 4 months.
3.2. Baseline Clinical Characteristics
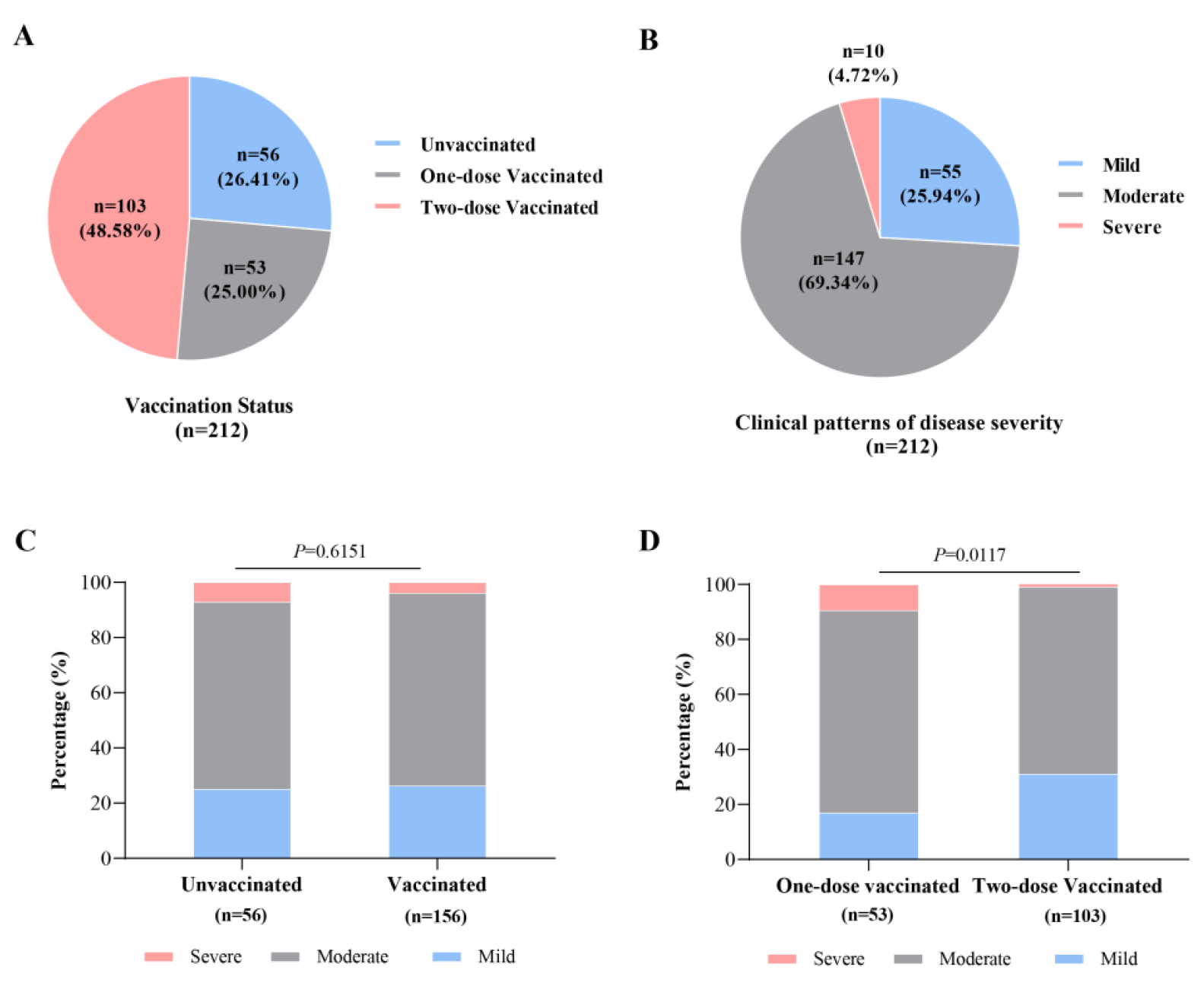
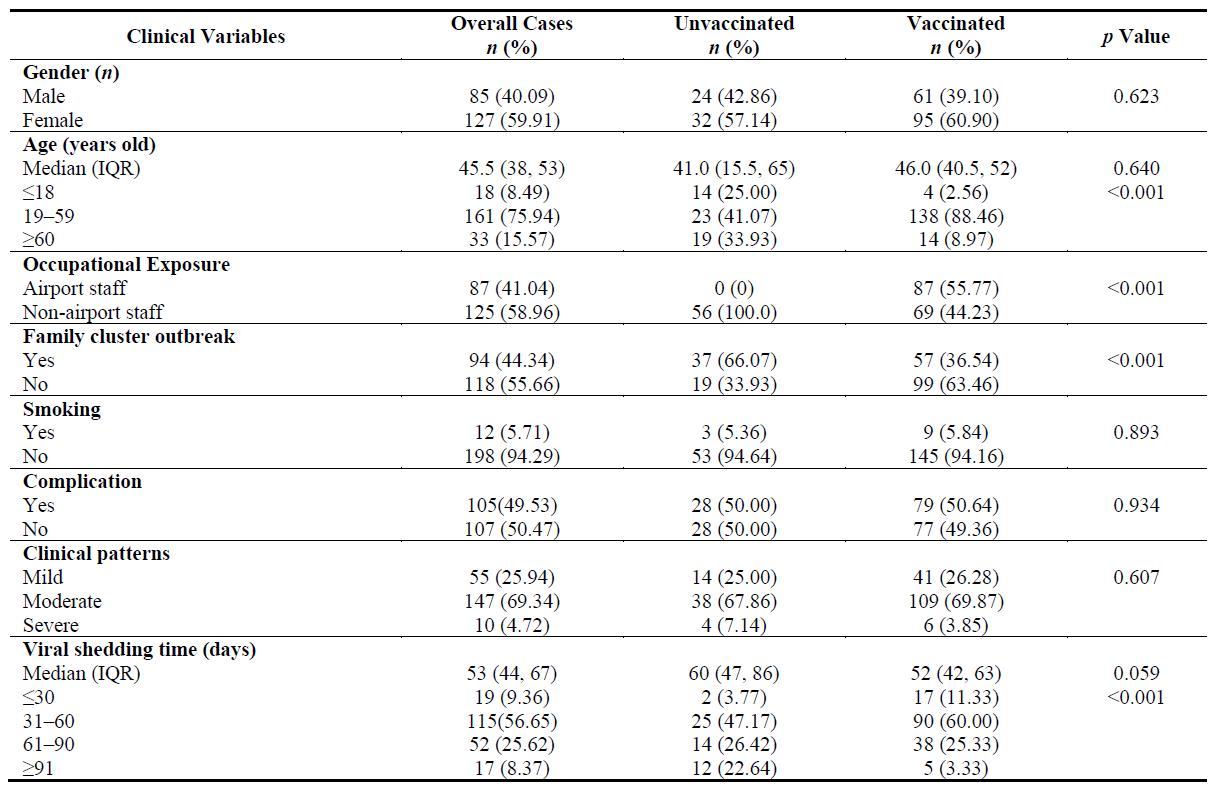
There were 212 patients with the COVID-19 Delta variant (unvaccinated, n = 56, 26.41%; one-dose vaccinated, n = 53, 25.00%; and two-dose vaccinated, n = 128, 48.58%) included in this cohort study. The median age was 45.5 (38, 53) years old, and 59.91% (n = 127) of the patients were women (Table 1 and Figure 3A). Eighty-seven cases (41.04%) were airport staff, and 32 families with 94 cases (44.34%) in the cluster were infected (average infected cases 3.1 per person) in the local outbreak. There were 55 (25.94%), 147 (69.34%) and 10 (4.72%) mild, moderate and severe cases, respectively (Table 1 and Figure 3B). The differences in age distribution, occupational exposure, family cluster outbreaks and VST were significant between the unvaccinated and vaccinated cases (p < 0.001, p < 0.001, p < 0.001 and p < 0.001, respectively). There were no significant differences in sex, smoking history, clinical disease severity, complications at different time points (p = 0.623, p = 0.893, p = 0.607 and p = 0.934, respectively) between the two groups of subjects (Table 1 and Figure 3C).
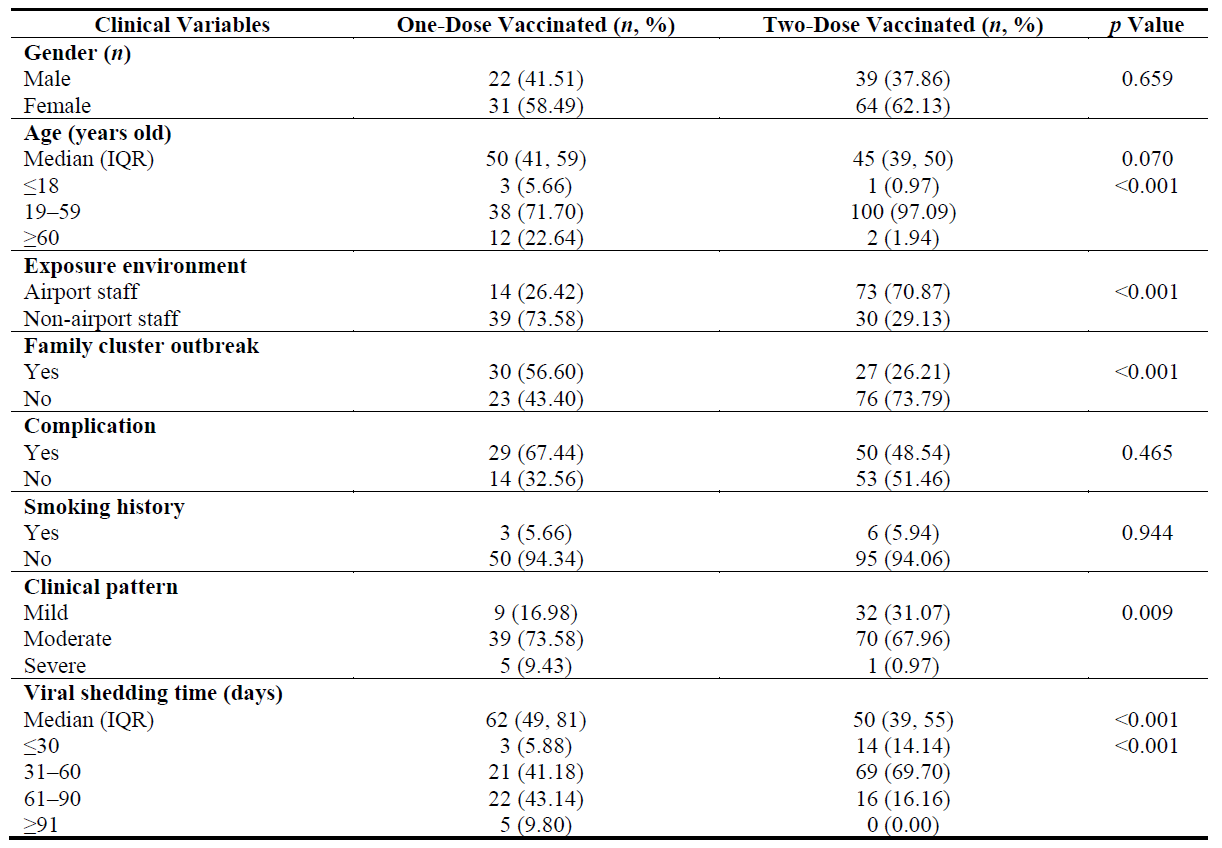
By comparing the clinical data of patients between the one-dose and two-dose vaccinated subgroups, the differences in age, occupational exposure, family cluster outbreaks, clinical disease severity and VST were significant (p < 0.001, p < 0.001, p < 0.001, p = 0.009 and p < 0.001, respectively) (Table 2). However, there were no significant differences in sex, complications or smoking history at the different time points (p = 0.659, p = 0.465 and p = 0.944, respectively) (Table 2 and Figure 3D).
The bivariate correlation analysis showed that the number of vaccine doses was negatively correlated with the clinical patterns (disease severity) in subjects with the COVID-19 Delta variant. Still, the difference was insignificant (r = −0.121, p = 0.078) (not shown).
3.3. The VST Can Be Considered the Period That an Individual Can Be Contagious
In our study, viral mRNA tests of 143 cases (67.45%) became positive again after an initial negative assessment. To accurately assess VST, there is a need to prolong the isolated observation period and increase the frequency of viral mRNA tests. The range of VST was from 20 to 132 days, and the median VST was 53 days in our cohort study.
The difference in VST was not significant between unvaccinated and vaccinated subjects (68.30 ± 26.24 vs. 53.26 ± 18.04 days, p = 0.0008) (Figure 4A). Still, the VST in one-dose vaccinated patients was significantly longer than that in two-dose vaccinated cases (63.61 ± 20.72 vs. 47.93 ± 13.83 days, p < 0.001) (Figure 4B). Significant differences in VST were observed among the three clinical pattern subgroups (p < 0.001), with significantly shorter VST in mild patients than in moderate cases (44.56 ± 15.81 vs. 59.96 ± 20.06 days, p < 0.0001) and significantly shorter VST in moderate patients than in severe cases (44.56 ± 15.81 vs. 93.63 ± 22.42 days, p = 0.0003) (Figure 4C). Additionally, VST was significantly longer in female and family cluster cases than in male and nonfamily cluster subjects (61.58 ± 22.03 vs. 50.29 ± 18.81 days, p = 0.0002 and 62.24 ± 24.27 vs. 52.92 ± 17.88, p = 0.0285, respectively) (Figure 4D,E). However, there were no significant differences in VST between airport and non-airport staff or among different age subgroups (p = 0.1282 and 0.1789, respectively) (Figure 4F,G).
3.4. The Predictive Factors for VST in Patients with the COVID-19 Delta Variant
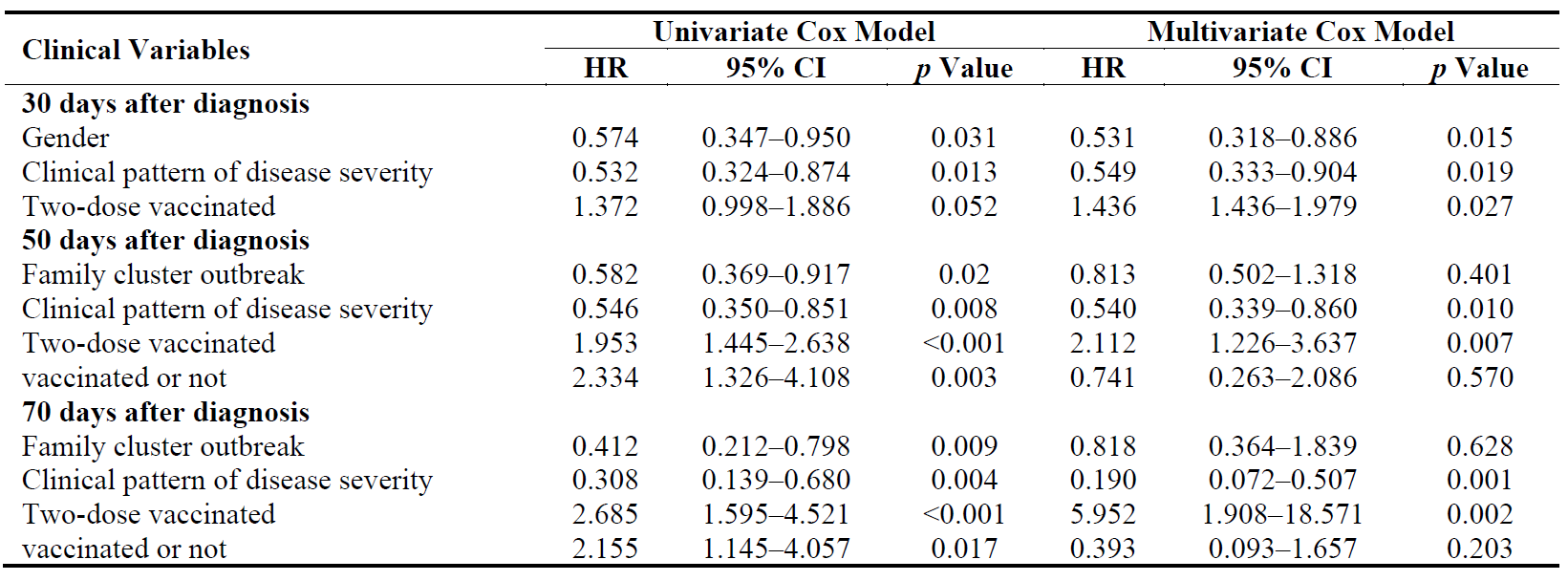
At 30 days after a COVID-19 Delta variant infection diagnosis, sex, clinical pattern of disease severity and vaccination dose were independent predictive factors for VST by multivariate Cox models. Family cluster, clinical pattern, vaccination dose and vaccination status were independent factors for VST according to multivariate Cox regression at 50 days after diagnosis. Additionally, at 70 days after diagnosis, family cluster, clinical pattern, vaccination dose, and vaccination status were also found to be associated with VST (Table 3).
From the above results, we found that clinical pattern of disease severity and vaccination dose were closely related to VST after diagnosis of COVID-19 Delta variant infection (Table 3).
Our data analysis revealed that several factors were independently associated with VST among individuals diagnosed with the COVID-19 Delta variant. Specifically, at 30 days after diagnosis, factors such as sex, clinical pattern of disease severity, and vaccination dose emerged as significant predictors of VST. Similarly, at 50 and 70 days after diagnosis, family cluster, clinical pattern, vaccination dose, and vaccination status were identified as independent factors associated with VST. These findings, as summarized in Table 3, highlight the importance of considering the clinical pattern of disease severity and the vaccination dose in relation to VST in individuals infected with the COVID-19 Delta variant.
Figure 2. <b>Time-series plots of patients with COVID-19 Delta variant in 2021 Summer Nanjing outbreak.</b> (<b>A</b>): The outbreak time of COVID-19 Delta. (<b>B</b>): The time of negative COVID-19 mRNA tests.
Figure 3. <b>The vaccination status and disease severity in 212 patients with COVID-19 Delta variant.</b> (<b>A</b>): The numbers of patients with COVID-19 Delta variant in different vaccination status. (<b>B</b>): The numbers of subjects with COVID-19 Delta variant in different disease severity. (<b>C</b>): Comparison of the percentages among cases with different disease severity between unvaccinated and vaccinated groups. (D): Comparison of percentages among cases with different disease severity between one-dose and two-dose vaccinated subgroups.
Table 1. Baseline clinical features of 212 patients with COVID-19 Delta variant.
Table 2. Baseline clinical characteristics in 156 vaccinated patients.
Figure 4. <b>Comparison of VST in different clinical subgroups.</b> (<b>A</b>): Between patients with vaccinated and unvaccinated patients (<i>p</i> < 0.0001. (<b>B</b>): Between one-dose and two-dose vaccinated subjects (<i>p</i> < 0.0001). (<b>C</b>): In patients with different disease severities (Between the Mild and Moderate, <i>p</i> < 0.0001; Between the Mild and Severe, <i>p</i> < 0.0001; Between the Moderate and Severe, <i>p</i> = 0.0003). (<b>D</b>): Between male and female subjects (<i>p</i> = 0.0002). (<b>E</b>): Between family and non-family cluster outbreak subjects (<i>p</i> = 0.0285). (<b>F</b>): Between airport staff and non-airport staff (<i>p</i> = 0.1282). (<b>G</b>): In different age subgroups (all <i>p</i> > 0.05).
Table 3. Cox regression analysis of the factors for VST in patients with COVID-19 Delta variant. HR, Hazard Ratio; 95% CI, 95% Confidence Interval.
4. Discussion
In our study, there were 212 patients with the COVID-19 Delta variant in the summer of 2021 during the Nanjing outbreak, including 56 unvaccinated, 53 one-dose vaccinated and 103 two-dose vaccinated cases. The VST was significantly shorter in the vaccinated subgroups, particularly those who had received two doses, and in the mild, male, and nonfamily cluster subgroups, compared to their counterparts. Moreover, it was observed that patients with mild symptoms had significantly shorter VSTs than those with moderate symptoms. In comparison, patients with moderate symptoms had significantly shorter VSTs than those with severe symptoms. Furthermore, Cox regression models indicated that both disease severity and vaccination dosage were independent predictors of VST. These findings suggest that inactivated vaccines manufactured in China may contribute towards reducing disease severity and that receiving two doses of the vaccine can help reduce VSTs in patients with the COVID-19 Delta variant.
Family clusters of infection have emerged as a major mode of COVID-19 transmission worldwide [16]. Within households, age and the presence of multiple comorbid conditions have been identified as risk factors associated with higher transmission rates [17]. The secondary attack rate of SARS-CoV-2 within households has been estimated at 16.3% [18]. Furthermore, completing the full vaccination schedule has been shown to reduce transmission of the Delta variant and lower the secondary attack rate among household contacts [19]. In our study, a significant proportion (44.34%) of cases were part of family cluster outbreaks involving 32 families with a total of 94 cases (an average of 2.94 infected cases per person in household transmission). Among the subjects in our study, 41.04% were airport staff. Notably, the majority of cases (95.28%) exhibited mild to moderate symptoms in our study. Some studies [15,20,21] have found that individuals infected with the COVID-19 Delta variant experienced a significantly decreased proportion of severe cases compared to those infected with the alpha variant, which contrasts with the symptoms commonly reported by alpha variant patients, such as fatigue, muscle weakness, sleep difficulties, and anxiety or depression, persisting even 6 months after acute infection. However, other studies ref. [22,23] have reported that Delta led to more severe disease than Alpha, while some [24] found no significant difference in disease severity between the two variants. Thus, the variability underscores the need for further research to clarify the impact of different variants on disease severity. A cohort study conducted in the UK revealed that fully vaccinated individuals who experienced breakthrough infections had peak viral loads similar to those of unvaccinated cases, suggesting the potential for transmission within their family [25]. Interestingly, the percentage of cases within family clustering outbreaks in our study was higher than that observed among household contacts in the UK. However, the differences in disease severity between the vaccinated and unvaccinated groups were not statistically significant, aligning with the findings from the UK study [25]. These findings suggest that while the COVID-19 Delta variant may exhibit higher infectiousness, it appears to be less virulent in terms of disease severity. This reinforces the importance of vaccination in mitigating the impact of the variant and preventing severe illness.
Low Ct values obtained from RT-PCR diagnostic tests have been linked to higher virus growth in cell culture, suggesting that individuals with lower Ct values can be infectious with prolonged viral RNA shedding [26]. Notably, a prospective cohort study showed that the Delta variant is associated with more severe symptoms and a longer duration of viral shedding compared to non-Delta variants [27]. A published study observed that individuals infected with the Delta variant who had received inactivated vaccines exhibited a shorter duration of viral shedding compared to unvaccinated individuals. Additionally, the clinical presentation of disease in vaccinated individuals was primarily asymptomatic or mild. The median duration of viral shedding in breakthrough infections was only 3 days, whereas it was 11 days in the unvaccinated group. Importantly, the duration of viral shedding in breakthrough infections was significantly shorter than in the unvaccinated group [28]. Adding to the growing body of evidence, an epidemiological investigation conducted in China found that the median shedding interval for the Delta variant was 12 days in asymptomatic/mild cases and 19 days in moderate/severe cases [29]. These studies highlight the complex interplay between vaccination, disease severity, and viral shedding dynamics in individuals infected with the Delta variant. Vaccination contributes to shorter viral shedding periods and helps reduce the severity of symptoms, emphasizing the importance of widespread vaccination efforts in controlling the spread of the Delta variant.
In our study, we observed a frequent reversion to positive results in COVID-19 Delta mRNA tests, with 67.45% of patients displaying a positive result after initially testing negative. This suggests that determining the exact duration of viral shedding requires longer observation and follow-up of these patients. To capture a more comprehensive picture of viral shedding, we extended our cohort's follow-up time for viral mRNA tests. Through repeated COVID-19 mRNA testing, we found that the VST range among the 212 patients infected with the COVID-19 Delta variant was between 20 and 132 days, with a median VST of 53 days. Interestingly, when comparing the unvaccinated and vaccinated groups, the median VST did not differ significantly (p = 0.059), but there was a notable difference in the distribution of VST (p < 0.001). Among vaccinated individuals, those who had received only one dose exhibited a significantly longer median VST (62 days, p < 0.001) compared to those who had received two doses (50 days). Furthermore, our analysis revealed a significant trend (p = 0.009), indicating a negative correlation between vaccination dose and disease severity in subjects infected with the COVID-19 Delta variant. This suggests that a higher vaccination dose may contribute to mitigating the severity of the disease. These findings emphasize the importance of considering the duration of viral shedding and the potential impact of vaccination in managing patients with the COVID-19 Delta variant. Prolonged viral shedding highlights the need for continued monitoring and adherence to infection control measures. At the same time, the correlation between vaccination dose and disease severity suggests the potential benefits of a robust vaccination strategy.
The effectiveness of COVID-19 vaccines in preventing infection and severe disease has been well-established, with two doses proving more effective than a single dose in countering the Delta variant [10]. Building upon this knowledge, our study specifically examined the impact of Chinese inactivated vaccines against COVID-19 on VST and disease severity in patients infected with the COVID-19 Delta variant.
The results from our analysis demonstrated that Chinese inactivated vaccines contributed to a reduction in VST among patients with the Delta variant, thereby indicating a more rapid clearance of the virus. Additionally, using these vaccines was associated with a mitigation of disease severity in affected individuals. This finding further emphasizes the beneficial effects of the vaccines in attenuating the impact of the Delta variant on patients’ health.
However, it is important to note that despite the positive impact of vaccination, the COVID-19 Delta variant is known to exhibit increased infectivity compared to previous variants, necessitating an extended duration of isolation for infected individuals. This highlights the ongoing challenges the Delta variant poses and underscores the need for continued preventive measures and vigilance in managing its spread.
A comprehensive report by the World Health Organization (WHO) revealed that over 166 COVID-19 vaccine candidates were in clinical development, with 22 of them being inactivated vaccines in the clinical phase [30]. Notably, a previous study demonstrated that a single dose of the vaccine provided approximately 80% effectiveness in preventing hospital admission and death specifically associated with the alpha variant [31]. Subsequent clinical trials further confirmed that the effectiveness of a single dose decreased to 30.7% against the Delta variant, while the effectiveness of two doses increased significantly to 88.0% [10]. Despite the high effectiveness of two doses against the Delta variant, there has been an increasing number of breakthrough infections reported in vaccinated individuals who became infected with the COVID-19 Delta variant. However, it is important to note that these breakthrough infections generally result in milder symptoms, and individuals with the Delta variant have exhibited lower rates of hospital admissions within 6 months of completing their vaccination regimen [32].
A comprehensive body of evidence from randomized controlled trials (RCTs) has consistently demonstrated the favorable attributes of inactivated SARS-CoV-2 vaccines. These studies have confirmed the vaccines’ excellent tolerability, safety profiles, and ability to induce robust humoral responses across all age groups. Importantly, the incidence of adverse reactions associated with these vaccines have remained low [11,33,34,35]. Although breakthrough infections involve many factors (the viral profile, immunity characteristics, host determinants and vaccination properties), the rates of breakthrough infection might be measured to indicate vaccine effectiveness [36,37]. A Chile trial showed that the rate of breakthrough cases was 1.99% in fully vaccinated subjects, and inadequate induction of neutralizing antibodies may be the major cause of breakthrough infections [38]. These findings indicate that the effectiveness of inactivated vaccines may decline after administering the second dose. Therefore, it is proposed that a third dose of the vaccine should be administered to enhance the levels of neutralizing antibodies [39,40]. This approach would offer individuals increased protection against COVID-19, thereby reinforcing the overall efficacy of the vaccination strategy.
The COVID-19 Delta variant with higher viral loads is remarkably transmissible, and the risk of disease progression is increased compared with the wild-type strain. The key questions for COVID-19 are the duration and determinants of infectious virus shedding. A study showed that quantitative viral RNA load assays could be used in test-based strategies to discontinue or de-escalate infection prevention and control precautions [41]. The prolonged viral RNA shedding predictors included sex, older age, comorbidities and initial viral load [42]. The trajectory of the SARS-CoV-2 viral load during the course of COVID-19 is relatively consistent, but the duration of infectivity remains uncertain [43]. Our study investigated various factors affecting VST at different time points following COVID-19 Delta variant infection. We identified sex, family cluster, disease severity, vaccination dose, and vaccination status as independent predictive factors. The median VST for the COVID-19 Delta variant was found to be 53 days. Consequently, the duration of COVID-19 Delta variant shedding may exceed that of other variants, warranting a minimum observation period of two months after diagnosis. These findings underscore the importance of prolonged monitoring and strict adherence to infection control measures, considering the extended shedding period of the COVID-19 Delta variant. It is crucial to remain vigilant and continue comprehensive surveillance to manage the transmission of this variant effectively.
Our study had several limitations that should be acknowledged. Firstly, it was a single-center cohort study, which may limit the generalizability of our findings to broader populations. Additionally, the number of samples included in our study was relatively small, which could have impacted our results' statistical power and precision. Secondly, we primarily relied on RT-PCR and Ct values to assess viral loading and shedding time. While these measures are widely used and informative, we did not assess other factors, such as neutralizing antibodies or the viability of the shed virus from the patients. These additional assessments could have provided a more comprehensive understanding of the immune response and the dynamics of viral shedding. Lastly, we were unable to analyze the specific effectiveness of different inactivated vaccines due to the limitations of the study population. Some patients were uncertain about the exact type of vaccine they had received, preventing us from conducting a robust comparative analysis. Moreover, it is important to acknowledge that the Delta variant is no longer the predominant strain, and current strains have evolved significantly. This limits the direct applicability of our findings to ongoing and future COVID-19 variants. The evolving nature of the virus underscores the need for continuous research to address new challenges and variants as they emerge.
5. Conclusions
In summary, Chinese inactivated vaccines could shorten the viral shedding time in patients with the COVID-19 Delta variant. These vaccines may decrease the disease severity of cases with the COVID-19 Delta variant. However, further exploration is needed in the future.
Supplementary Materials
The following supporting information can be found at: https://www.sciepublish.com/article/pii/290, Figure S1. The transfer flow chart of patients with COVID-19 Delta variant.
Acknowledgments
The authors would like to thank all patients for sending their consents.
Author Contributions
M.C., F.L. (Fuqun Liu) and S.W. conceived and prepared the manuscript. M.C., F.L. (Fuqun Liu), L.W., F.L. (Fuchao Li), W.W. (Weihua Wu), Y.X. (Ying Xu), Z.Z., Y.X. (Yangyang Xia), Y.W., Y.Y., W.W. (Wei Wang) and H.Z. administrated these patients. M.C., F.L. (Fuqun Liu), L.W., M.L. and F.L. (Fuchao Li) collected the clinical data. M.C., F.L. (Fuqun Liu), S.W. and X.H. contributed to analysis of data, make the statistics and draft the manuscript. M.C., S.W. and H.Z. reviewed the manuscript and took responsibility for the integrity and accuracy of data analysis. All authors reviewed and approved the final draft.
Ethics Statement
The studies involving human participants were reviewed and approved by Ethics Committee of Lishui District Traditional Hospital of Chinese Medicine according with the Declaration of Helsinki (1989) (NO. 2021LCGC001). The patients/participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Funding
This study was partly supported by National Natural Science Foundation of China (82070064, 81670059 and 81200049), Fundings for Clinical Trials from the Affiliated Drum Tower Hospital, Medical School of Nanjing University (2022-LCYJ-MS-11) and the Fifth Batch of Chinese Medicine Emergency Special Fund Project for Pneumonia in Novel Coronavirus, National Administration of Traditional Chinese Medicine in 2021 (2021ZYLCYJ05-3).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding authors.
References
1.
WHO. COVID-19 Epidemiological Update, Edition 170, 13 August 2024; World Health Organization: Geneva, Switzerland, 2024.
2.
Cevik M, Grubaugh ND, Iwasaki A, Openshaw P. COVID-19 vaccines: Keeping pace with SARS-CoV-2 variants. Cell 2021, 184, 5077–5081. [Google Scholar]
3.
Callaway E, Ledford H. How bad is Omicron? What scientists know so far. Nature 2021, 600, 197–199. [Google Scholar]
4.
Ferreira I, Kemp SA, Datir R, Saito A, Meng B, Rakshit P, et al. SARS-CoV-2 B.1.617 Mutations L452R and E484Q Are Not Synergistic for Antibody Evasion. J. Infect. Dis. 2021, 224, 989–994. [Google Scholar]
5.
Ong SWX, Chiew CJ, Ang LW, Mak TM, Cui L, Toh MPHS, et al. Clinical and Virological Features of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Variants of Concern: A Retrospective Cohort Study Comparing B.1.1.7 (Alpha), B.1.351 (Beta), and B.1.617.2 (Delta). Clin. Infect. Dis. 2022, 75, e1128–e1136. [Google Scholar]
6.
Baay M, Lina B, Fontanet A, Marchant A, Saville M, Sabot P, et al. SARS-CoV-2: Virology, epidemiology, immunology and vaccine development. Biologicals 2020, 66, 35–40. [Google Scholar]
7.
Mlcochova P, Kemp SA, Dhar MS, Papa G, Meng B, Ferreira IATM, et al. SARS-CoV-2 B.1.617.2 Delta variant replication and immune evasion. Nature 2021, 599, 114–119. [Google Scholar]
8.
Twohig KA, Nyberg T, Zaidi A, Thelwall S, Sinnathamby MA, Aliabadi S, et al. Hospital admission and emergency care attendance risk for SARS-CoV-2 delta (B.1.617.2) compared with alpha (B.1.1.7) variants of concern: A cohort study. Lancet Infect. Dis. 2022, 22, 35–42. [Google Scholar]
9.
Feikin DR, Higdon MM, Abu-Raddad LJ, Andrews N, Araos R, Goldberg Y, et al. Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: Results of a systematic review and meta-regression. Lancet 2022, 399, 924–944. [Google Scholar]
10.
Lopez Bernal J, Andrews N, Gower C, Gallagher E, Simmons R, Thelwall S, et al. Effectiveness of COVID-19 Vaccines against the B.1.617.2 (Delta) Variant. N. Engl. J. Med. 2021, 385, 585–594. [Google Scholar]
11.
Zhang Y, Zeng G, Pan H, Li C, Hu Y, Chu K, et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18–59 years: A randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect. Dis. 2021, 21, 181–192. [Google Scholar]
12.
Al Kaabi N, Zhang Y, Xia S, Yang Y, Al Qahtani MM, Abdulrazzaq N, et al. Effect of 2 Inactivated SARS-CoV-2 Vaccines on Symptomatic COVID-19 Infection in Adults: A Randomized Clinical Trial. JAMA 2021, 326, 35–45. [Google Scholar]
13.
Tanriover MD, Doğanay HL, Akova M, Güner HR, Azap A, Akhan S, et al. Efficacy and safety of an inactivated whole-virion SARS-CoV-2 vaccine (CoronaVac): Interim results of a double-blind, randomised, placebo-controlled, phase 3 trial in Turkey. Lancet 2021, 398, 213–222. [Google Scholar]
14.
Liu F, Zheng C, Wang L, Geng M, Chen H, Zhou S, et al. Interpretation of the Protocol for Prevention and Control of COVID-19 in China (Edition 8). China CDC Wkly. 2021, 3, 527–530. [Google Scholar]
15.
Li XN, Huang Y, Wang W, Jing QL, Zhang CH, Qin PZ, et al. Effectiveness of inactivated SARS-CoV-2 vaccines against the Delta variant infection in Guangzhou: A test-negative case-control real-world study. Emerg. Microbes Infect. 2021, 10, 1751–1759. [Google Scholar]
16.
Thompson HA, Mousa A, Dighe A, Fu H, Arnedo-Pena A, Barrett P, et al. Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Setting-specific Transmission Rates: A Systematic Review and Meta-analysis. Clin. Infect. Dis. 2021, 73, e754–e764. [Google Scholar]
17.
Metlay JP, Haas JS, Soltoff AE, Armstrong KA. Household Transmission of SARS-CoV-2. JAMA Netw. Open 2021, 4, e210304. [Google Scholar]
18.
Li W, Zhang B, Lu J, Liu S, Chang Z, Peng C, et al. Characteristics of Household Transmission of COVID-19. Clin. Infect. Dis. 2020, 71, 1943–1946. [Google Scholar]
19.
de Gier B, Andeweg S, Joosten R, Ter Schegget R, Smorenburg N, et al. Vaccine effectiveness against SARS-CoV-2 transmission and infections among household and other close contacts of confirmed cases, the Netherlands, February to May 2021. Eurosurveillance 2021, 26, 2100640. [Google Scholar]
20.
Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar]
21.
Huang C, Huang L, Wang Y, Li X, Ren L, Gu X, et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar]
22.
Varea-Jiménez E, Aznar Cano E, Vega-Piris L, Martínez Sánchez EV, Mazagatos C, García San Miguel Rodríguez-Alarcón L, et al. Comparative severity of COVID-19 cases caused by Alpha, Delta or Omicron SARS-CoV-2 variants and its association with vaccination. Enfermedades Infecc. Y Microbiol. Clin. 2023, 42, 187–194. (In English)[Google Scholar]
23.
Pascall DJ, Vink E, Blacow R, Bulteel N, Campbell A, Campbell R, et al. Directions of change in intrinsic case severity across successive SARS-CoV-2 variant waves have been inconsistent. J. Infect. 2023, 87, 128–135. [Google Scholar]
24.
Esper FP, Adhikari TM, Tu ZJ, Cheng YW, El-Haddad K, Farkas DH, et al. Alpha to Omicron: Disease Severity and Clinical Outcomes of Major SARS-CoV-2 Variants. J. Infect. Dis. 2022, 227, 344–352. [Google Scholar]
25.
Singanayagam A, Hakki S, Dunning J, Madon KJ, Crone MA, Koycheva A, et al. Community transmission and viral load kinetics of the SARS-CoV-2 delta (B.1.617.2) variant in vaccinated and unvaccinated individuals in the UK: A prospective, longitudinal, cohort study. Lancet Infect. Dis. 2022, 22, 183–195. [Google Scholar]
26.
Gniazdowski V, Paul Morris C, Wohl S, Mehoke T, Ramakrishnan S, Thielen P, et al. Repeated Coronavirus Disease 2019 Molecular Testing: Correlation of Severe Acute Respiratory Syndrome Coronavirus 2 Culture With Molecular Assays and Cycle Thresholds. Clin. Infect. Dis. 2021, 73, e860–e869. [Google Scholar]
27.
Park S, Lim SY, Kim JY, Park H, Lim JS, Bae S, et al. Clinical and Virological Characteristics of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) B.1.617.2 (Delta) Variant: A Prospective Cohort Study. Clin. Infect. Dis. 2022, 75, e27–e34. [Google Scholar]
28.
Tian D, Song Y, Zhang M, Pan Y, Ge Z, Zhang Y, et al. Genomic, immunological, and clinical analysis of COVID-19 vaccine breakthrough infections in Beijing, China. J. Med. Virol. 2022, 94, 2237–2249. [Google Scholar]
29.
Li B, Deng A, Li K, Hu Y, Li Z, Shi Y, et al. Viral infection and transmission in a large, well-traced outbreak caused by the SARS-CoV-2 Delta variant. Nat. Commun. 2022, 13, 460. [Google Scholar]
30.
World Health Organization Draft Landscape of COVID-19 Candidate Vaccines. Available online: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines (accessed on 30 March 2023).
31.
Lopez Bernal J, Andrews N, Gower C, Robertson C, Stowe J, Tessier E, et al. Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca vaccines on COVID-19 related symptoms, hospital admissions, and mortality in older adults in England: Test negative case-control study. BMJ 2021, 373, n1088. [Google Scholar]
32.
Tartof SY, Slezak JM, Fischer H, Hong V, Ackerson BK, Ranasinghe ON, et al. Effectiveness of mRNA BNT162b2 COVID-19 vaccine up to 6 months in a large integrated health system in the USA: A retrospective cohort study. Lancet 2021, 398, 1407–1416. [Google Scholar]
33.
Han B, Song Y, Li C, Yang W, Ma Q, Jiang Z, et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine (CoronaVac) in healthy children and adolescents: A double-blind, randomised, controlled, phase 1/2 clinical trial. Lancet Infect. Dis. 2021, 21, 1645–1653. [Google Scholar]
34.
Wu Z, Hu Y, Xu M, Chen Z, Yang W, Jiang Z, et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine (CoronaVac) in healthy adults aged 60 years and older: A randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect. Dis. 2021, 21, 803–812. [Google Scholar]
35.
Zhang MX, Zhang TT, Shi GF, Cheng FM, Zheng YM, Tung TH, et al. Safety of an inactivated SARS-CoV-2 vaccine among healthcare workers in China. Expert. Rev. Vaccines 2021, 20, 891–898. [Google Scholar]
36.
Amanatidou E, Gkiouliava A, Pella E, Serafidi M, Tsilingiris D, Vallianou NG, et al. Breakthrough infections after COVID-19 vaccination: Insights, perspectives and challenges. Metabol. Open 2022, 14, 100180. [Google Scholar]
37.
Siedner MJ, Boucau J, Gilbert RF, Uddin R, Luu J, Haneuse S, et al. Duration of viral shedding and culture positivity with postvaccination SARS-CoV-2 delta variant infections. JCI Insight 2022, 7, e155483. [Google Scholar]
38.
Duarte LF, Gálvez NMS, Iturriaga C, Melo-González F, Soto JA, Schultz BM, et al. Immune Profile and Clinical Outcome of Breakthrough Cases After Vaccination With an Inactivated SARS-CoV-2 Vaccine. Front. Immunol. 2021, 12, 742914. [Google Scholar]
39.
Zeng G, Wu Q, Pan H, Li M, Yang J, Wang L, et al. Immunogenicity and safety of a third dose of CoronaVac, and immune persistence of a two-dose schedule, in healthy adults: Interim results from two single-centre, double-blind, randomised, placebo-controlled phase 2 clinical trials. Lancet Infect. Dis. 2022, 22, 483–495. [Google Scholar]
40.
Wang K, Cao Y, Zhou Y, Wu J, Jia Z, Hu Y, et al. A third dose of inactivated vaccine augments the potency, breadth,and duration of anamnestic responses against SARS-CoV-2. medRxiv 2021, doi: 10.1101/2021.09.02.21261735.
41.
van Kampen JJA, van de Vijver D, Fraaij PLA, Haagmans BL, Lamers MM, Okba N, et al. Duration and key determinants of infectious virus shedding in hospitalized patients with coronavirus disease-2019 (COVID-19). Nat. Commun. 2021, 12, 267. [Google Scholar]
42.
Chen Y, Klein SL, Garibaldi BT, Li H, Wu C, Osevala NM, et al. Aging in COVID-19: Vulnerability, immunity and intervention. Ageing Res. Rev. 2021, 65, 101205. [Google Scholar]
43.
Walsh KA, Jordan K, Clyne B, Rohde D, Drummond L, Byrne P, et al. SARS-CoV-2 detection, viral load and infectivity over the course of an infection. J. Infect. 2020, 81, 357–371. [Google Scholar]