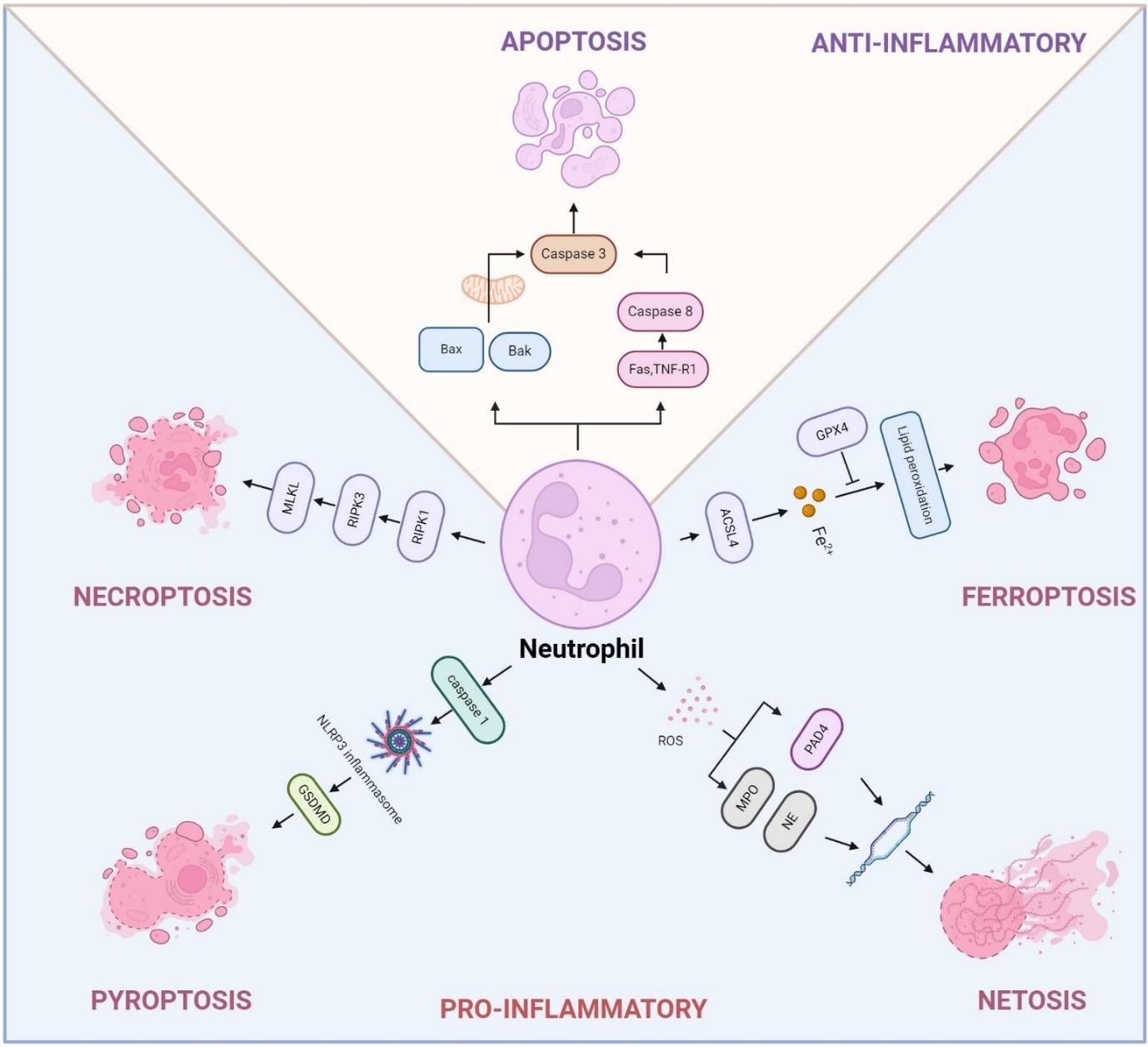
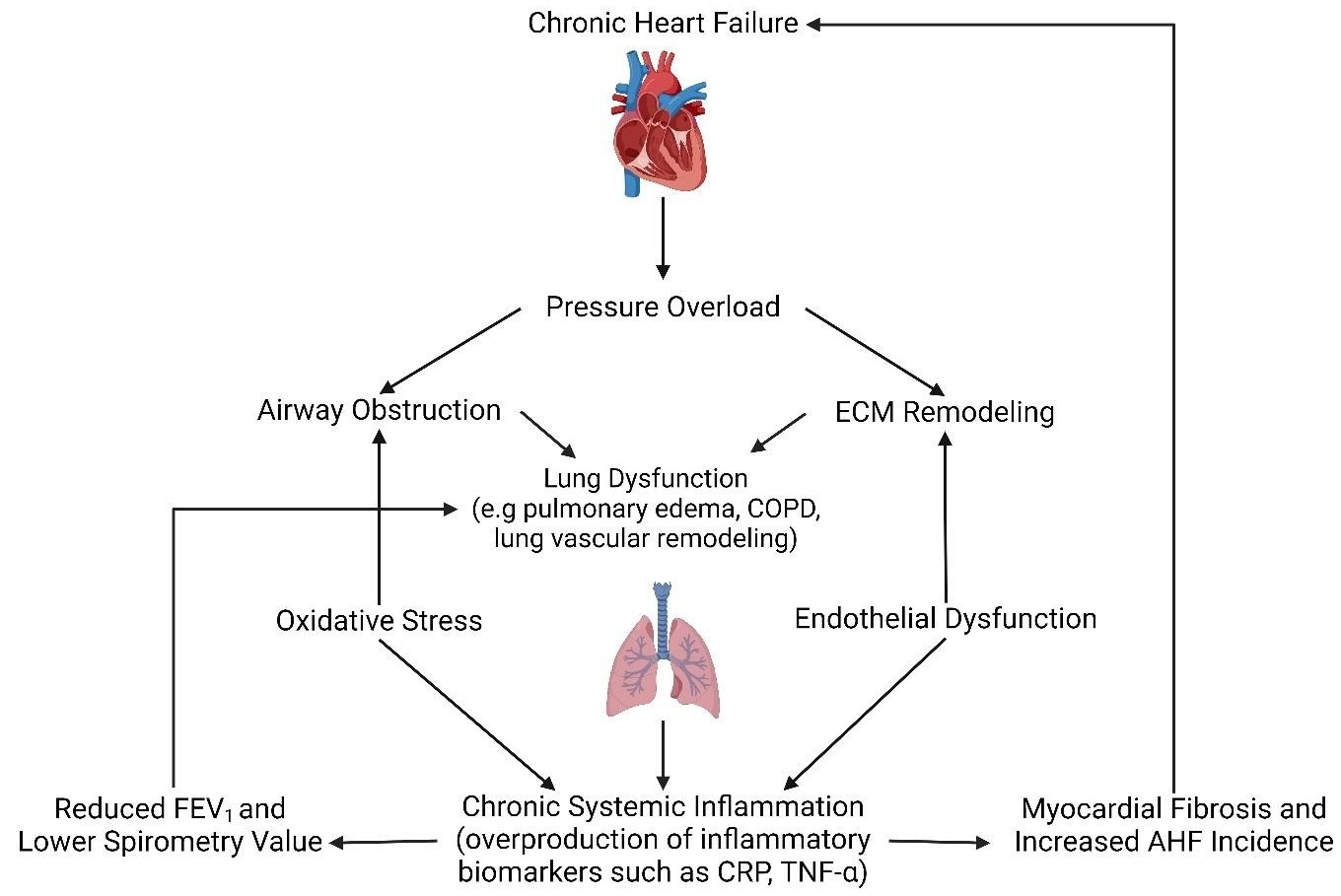
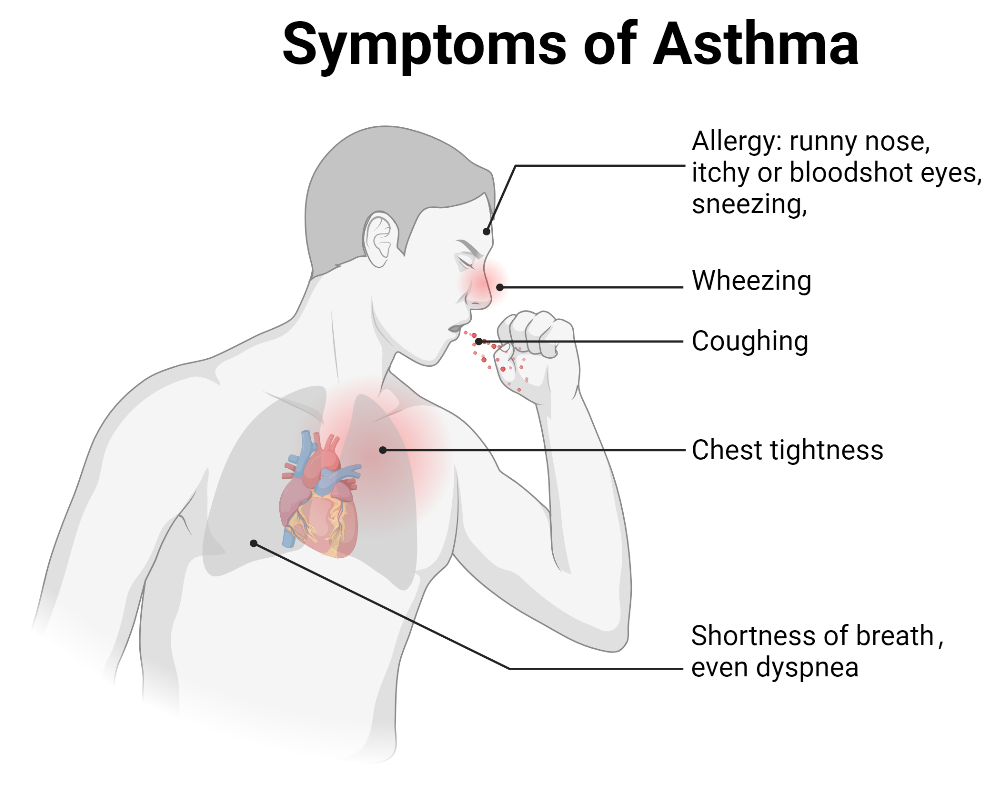
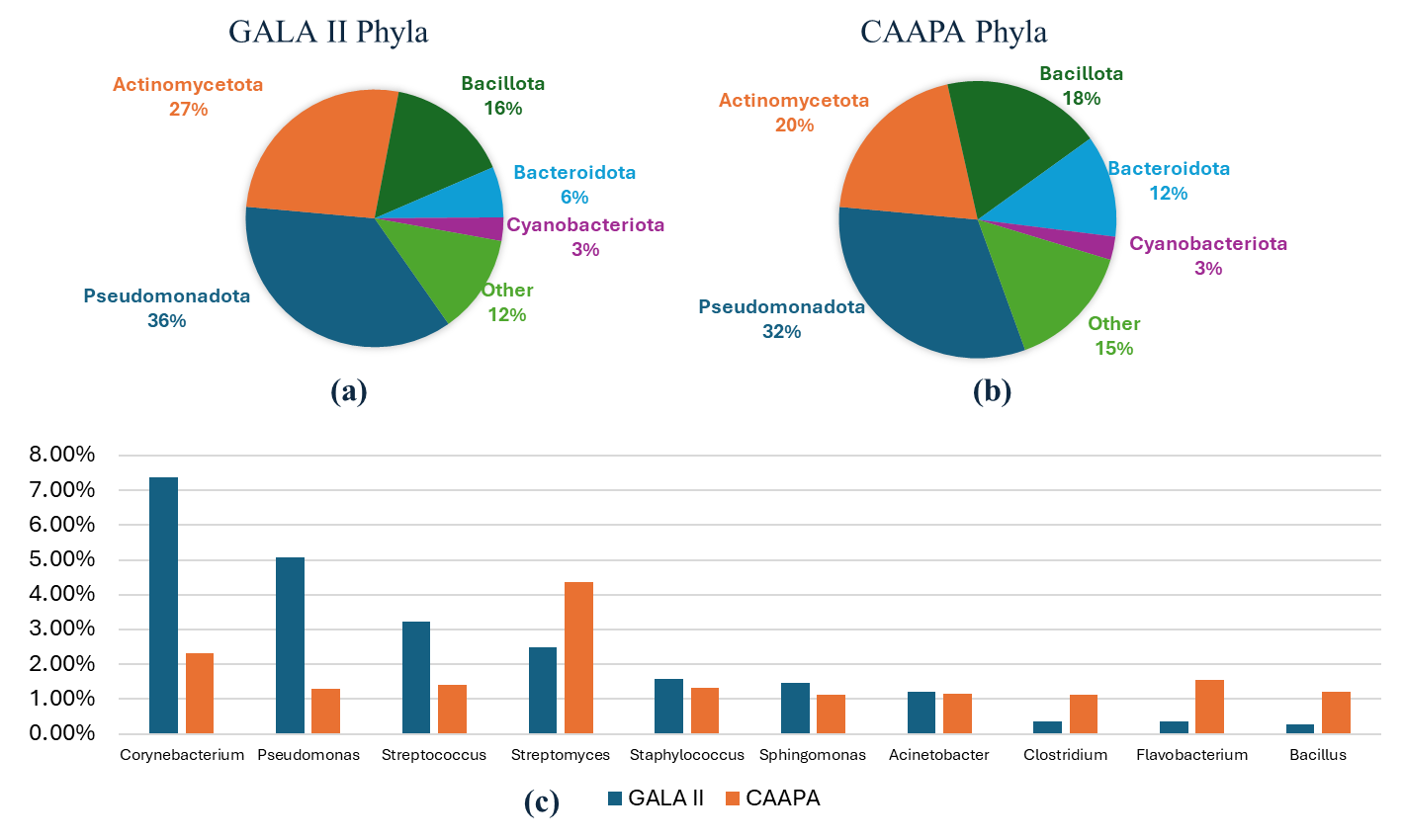
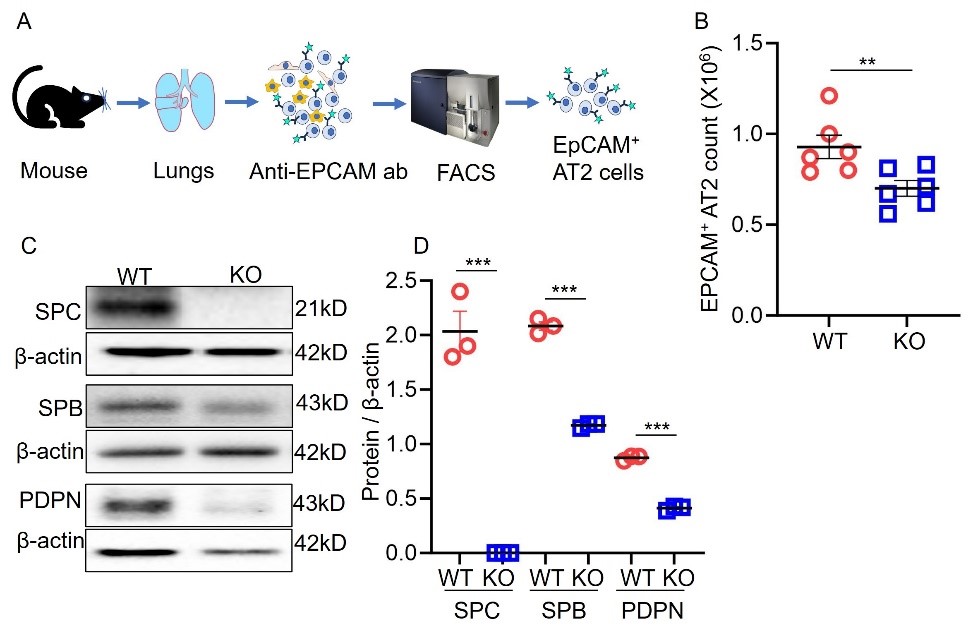
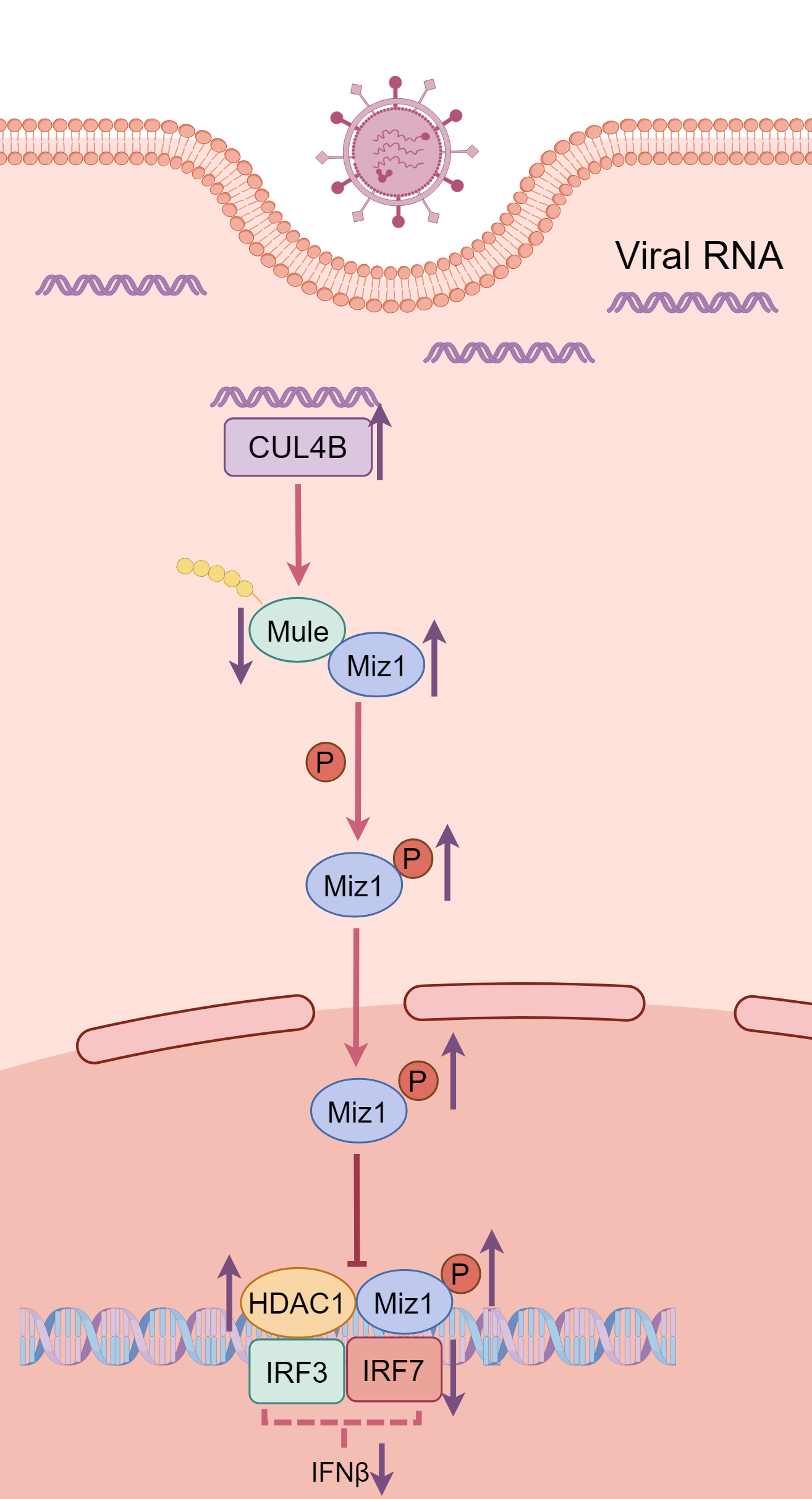
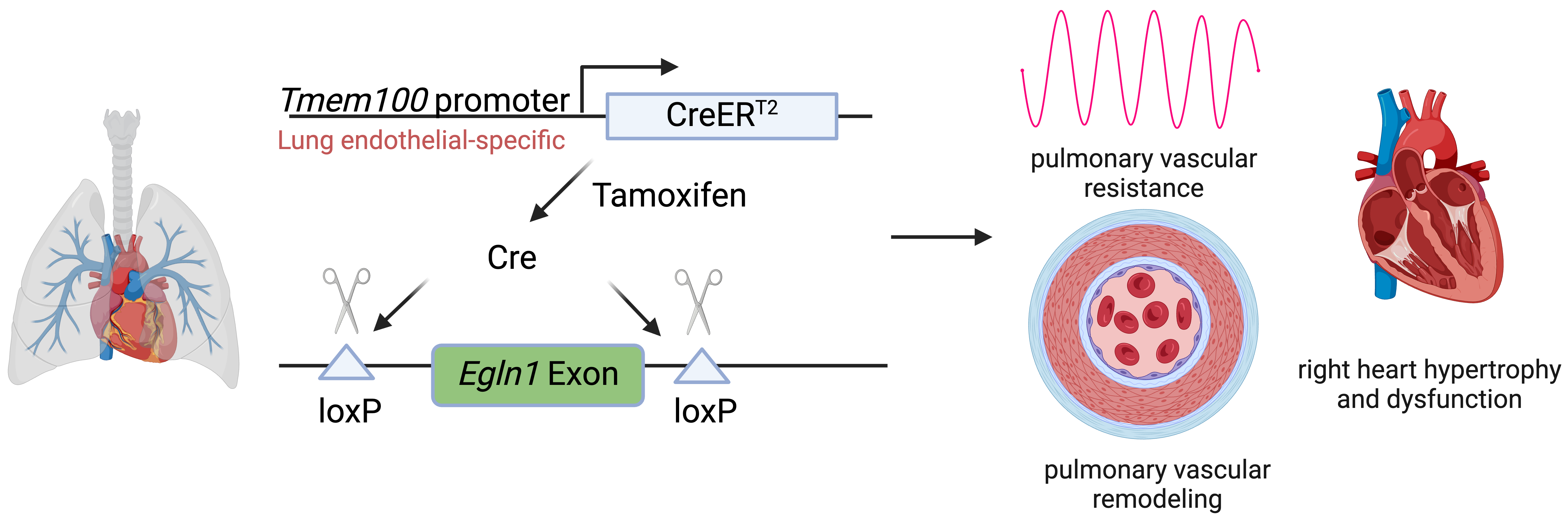
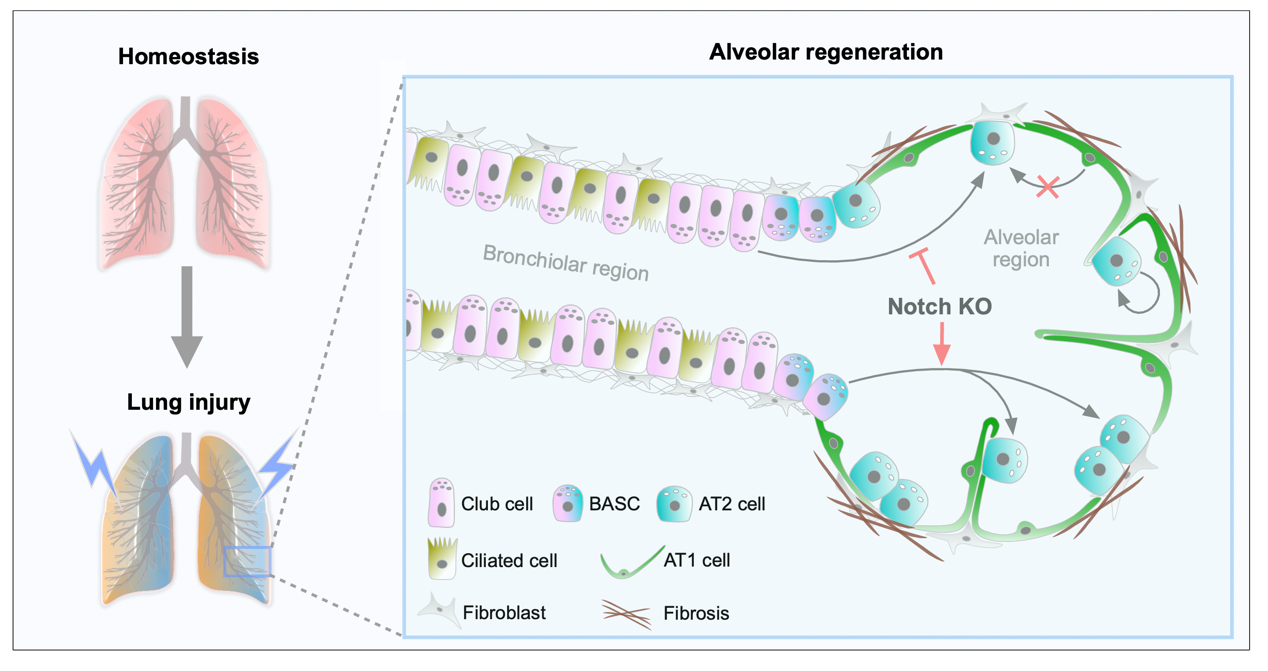
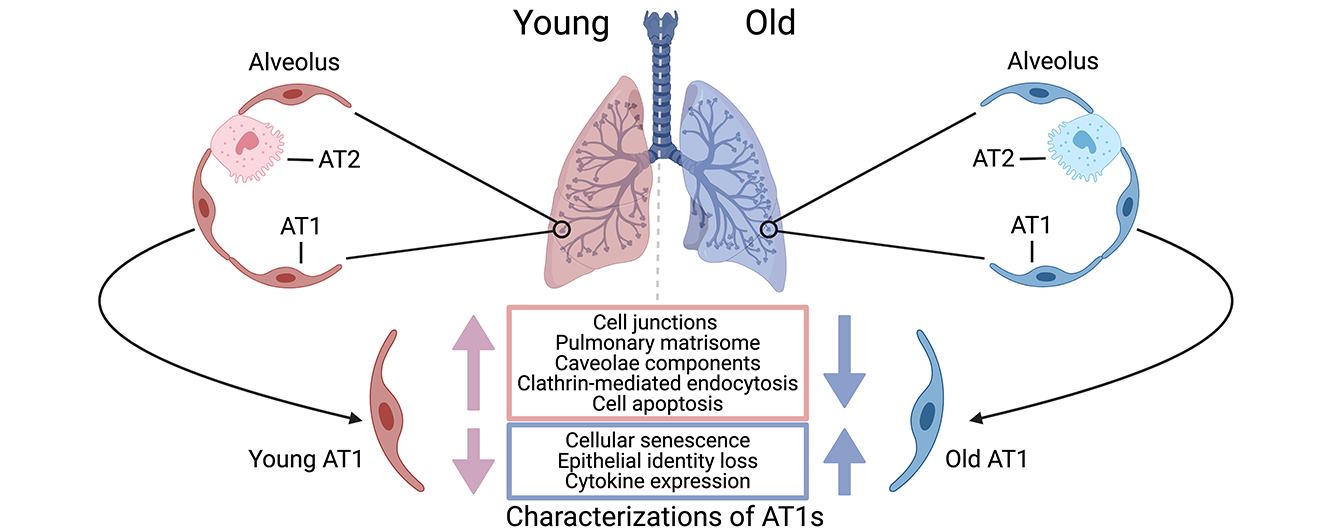
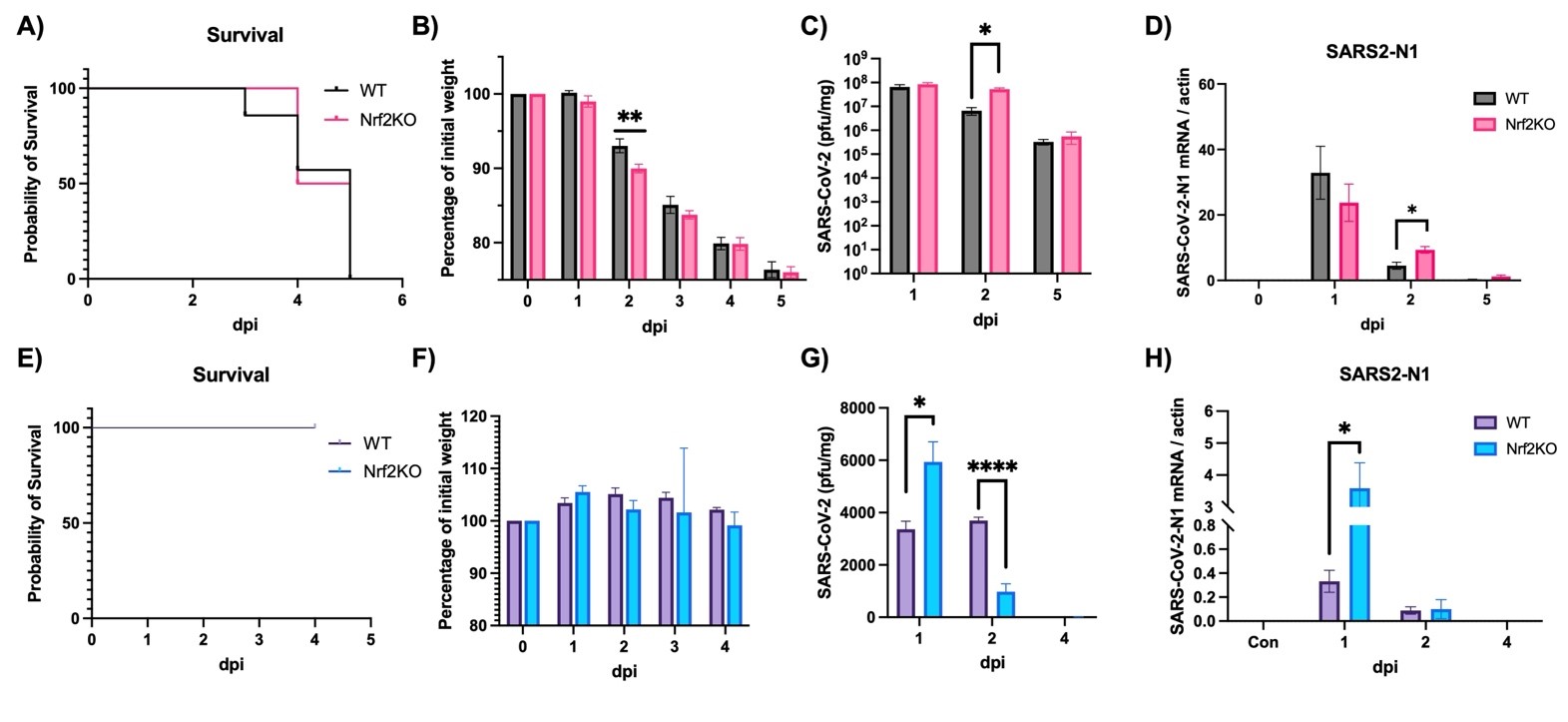
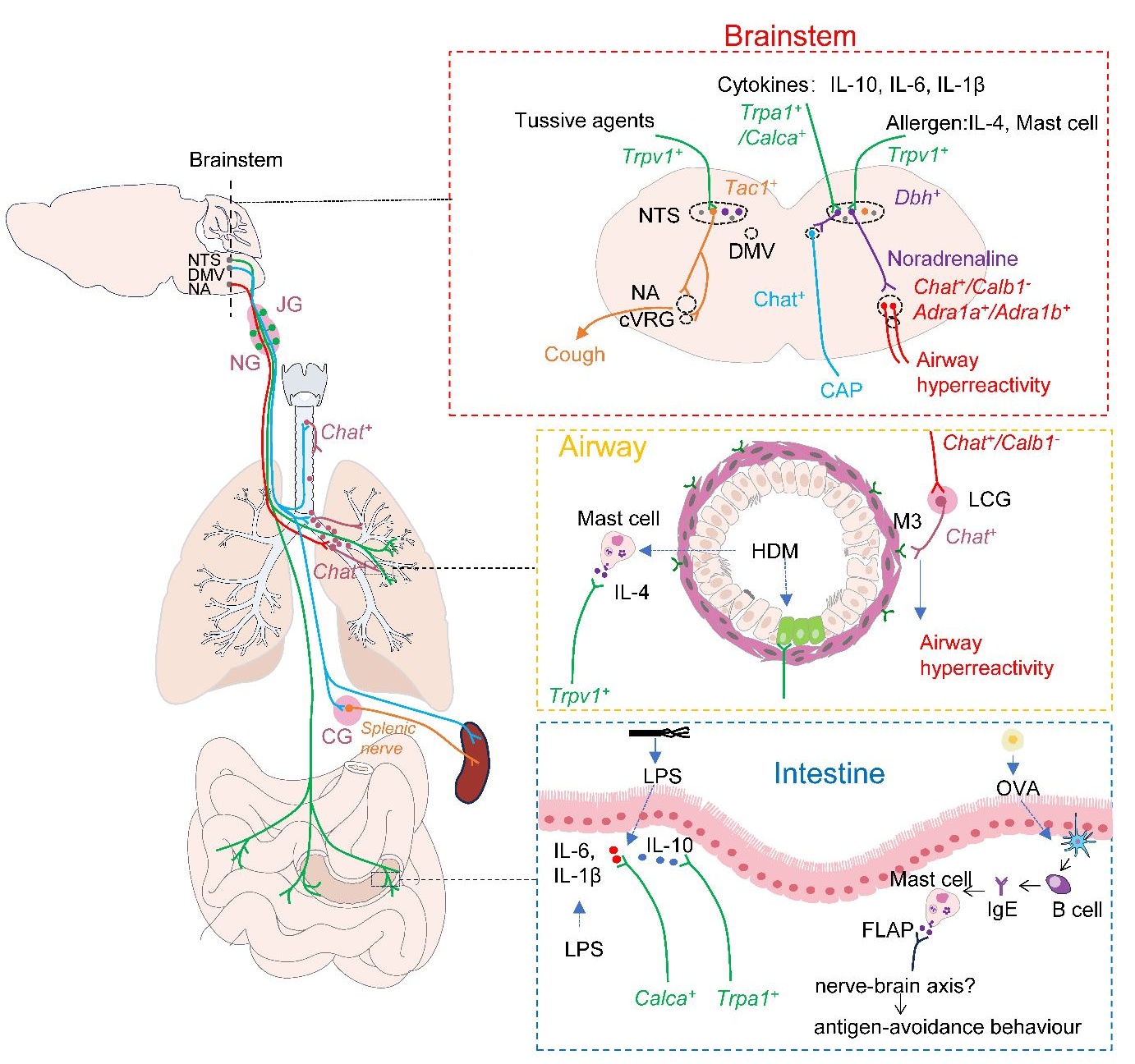
Journal of Respiratory Biology and Translational Medicine
 Open Access
Open Access
ISSN: 3006-6514 (Online)
3006-6506 (Print)
Journal of Respiratory Biology and Translational Medicine is an open access, peer-reviewed online journal that publishes basic, clinical, and translational lung and respiratory research. It is published quarterly online by SCIEPublish.