Found 5 results
Article
21 February 2025Efficient Removal of Glyphosate from Aqueous Solution by Cerium Dioxide Loaded Biochar
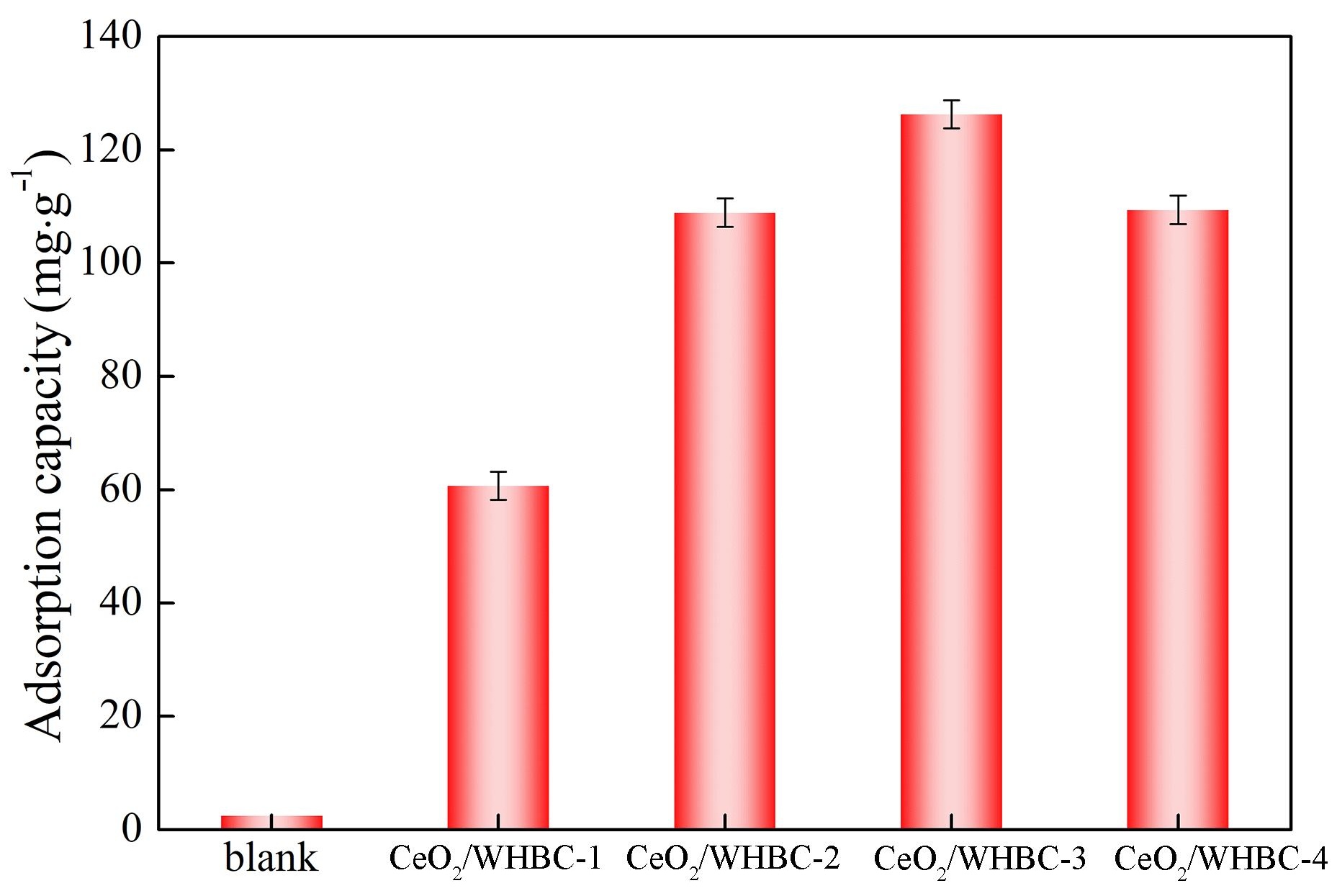
Glyphosate, which is one of the most widely used organophosphorus herbicides, poses a threat to the surrounding water environment. Traditional adsorbents were depicted to have poor capacities to eliminate it. CeO2 embraces the potential to adsorb glyphosate efficiently. However, suitable carbonaceous composites were necessary to be employed as its support. In this paper, water hyacinth was used as the precursor to prepare CeO2-loaded biochar (CeO2/WHBC), which was employed to remove glyphosate from the aqueous solution via adsorption. The results showed that CeO2/WHBC-3 illustrated the best adsorption performance for glyphosate with the capacity of 126.3 mg·g, which was prepared with per mmol CeO2 loaded of 0.2 g WHCB. Static adsorption experiments demonstrated that glyphosate adsorption at different solution pH values followed the Langmuir isotherm model and quasi-second order kinetic model, indicating that the adsorption was monolayer adsorption and that the adsorbent’s surface active sites primarily controlled the rate. Coexisting ion interference experiments showed that common cations (K+, Na+, Ca2+, Mg2+) and anions (Cl−, NO3−, SO42−) both promoted glyphosate adsorption on the CeO2/WHBC-3 surface. Moreover, the prepared sorbent maintained a high adsorption capacity after five adsorption-desorption cycles. Dynamic adsorption experiments showed that the CeO2/WHBC-3 packed column could efficiently remove glyphosate from aqueous solutions, even at high concentrations and fast flow rates. Zeta potentials and XPS analysis revealed that the adsorption mechanism of CeO2/WHBC-3 for glyphosate is mainly through electrostatic adsorption and metal complexation.
Review
20 January 2025Adsorption and High-Value Transformation of Volatile Fatty Acids from Microbial Fermentation Products: A Review
To mitigate the aforementioned global environmental issues, the concept of carbon capture and storage is crucial in addressing the necessity for carbon peaking and carbon neutrality. The buildup of volatile fatty acids during anaerobic fermentation is a primary factor contributing to the suboptimal performance or outright failure of anaerobic digestion systems. In response to the pressing demand for volatile organic acid recovery and high-value conversion, we primarily outlined the sources, recovery techniques, adsorption materials, and methods for high-value conversion of volatile fatty acids. The methods of adsorbing volatile acetic acid were presented, encompassing adsorption materials, mechanisms, and interfacial modifications of the adsorbent. Furthermore, drawing from recent research advancements, we have synthesized the high-value conversion techniques for volatile fatty acids and evaluated the research challenges and future prospects in this domain.
Article
12 December 2024Adsorption of Bisphenol A and 2,6-Dichlorophenol in Water Using Magnetic Phosphogypsum Composite Materials
Phenolic pollutants in water bodies pose a huge threat to human health and environmental safety. In this paper, a hydrophobicity-enhanced magnetic C-SiO2/MPG composite was prepared by a two-step method to remove bisphenol A (BPA)and 2,6-dichlorophenol (2,6-DCP), typical phenolic trace pollutants in livestock wastewater and natural water bodies. The results of pH gradient experiments showed that C-SiO2/MPG showed a stable removal effect on BPA in the pH range of 2–11. The adsorption of 2,6-DCP by C-SiO2/MPG peaked at pH = 2, while the adsorption of 2,6-DCP by C-SiO2/MPG was severely inhibited under alkaline conditions. The PSO kinetic model and the Langmuir isotherm model can better describe the adsorption process of BPA and 2,6-DCP on C-SiO2/MPG, indicating that the monolayer chemical adsorption has a rate-controlling step. With the Langmuir equation fitting, the maximum adsorption capacity of C-SiO2/MPG for BPA and 2,6-DCP at 298 K was calculated to be 561.79 mg/g and 531.91 mg/g, respectively. The results of adsorption thermodynamics indicated that the adsorption of BPA and 2,6-DCP on C-SiO2/MPG was spontaneous, accompanied by a process of entropy decrease. C-SiO2/MPG showed good environmental resistance and repeated use stability for BPA and 2,6-DCP in electrolyte ion interference, actual water samples and cycle experiments. Mechanism analysis showed that the adsorption of BPA and 2,6-DCP on C-SiO2/MPG was mainly controlled by hydrogen bonding and hydrophobic interactions. This study designed an efficient adsorbent for phenolic pollutants that can be used in actual wastewater and broadened the resource utilization of industrial waste phosphogypsm.

Article
14 November 2024The Discovering of Rapid Formation La(OH)3 from LaH3
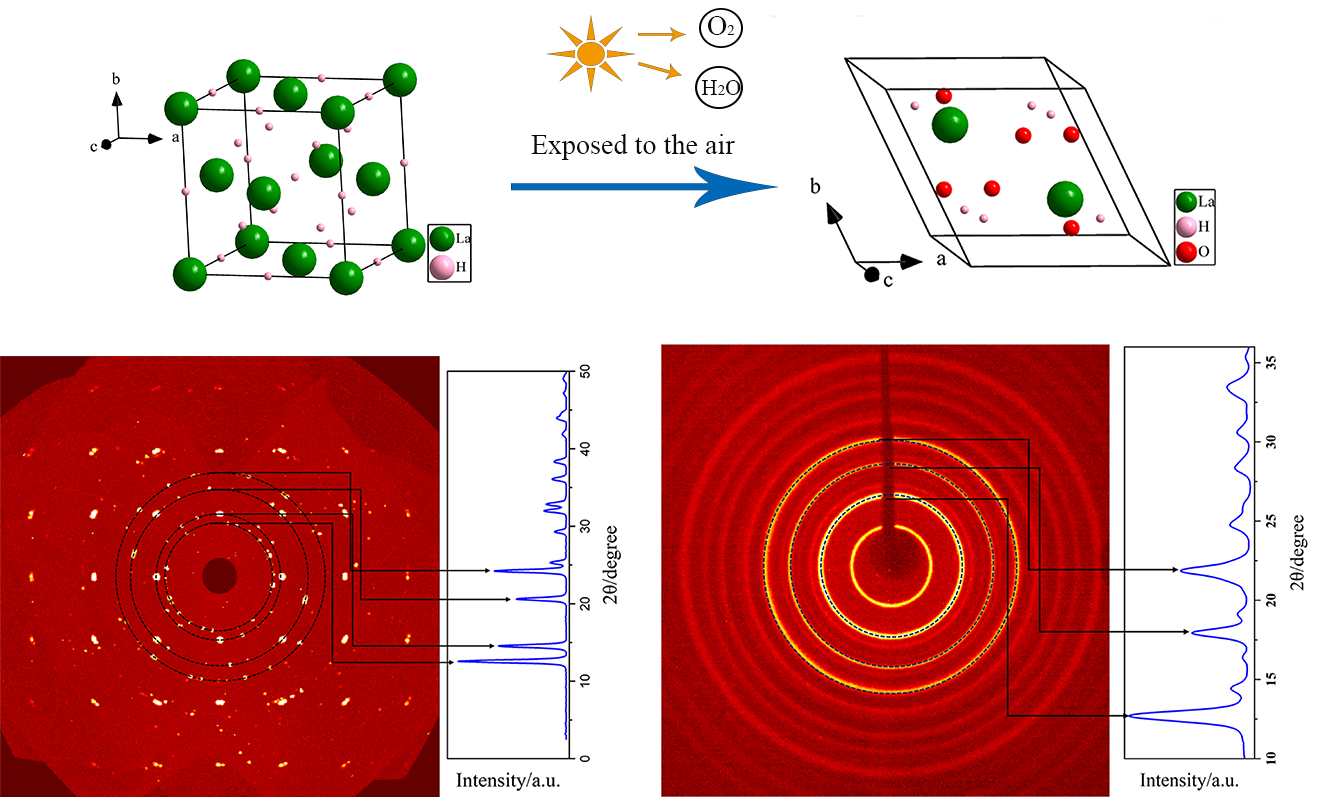
It was found that the single crystal of LaH3 specimen with $${Fm\overline{3}m}$$ (No.225) will decompose into powders within 24 h, which is later characterized to be La(OH)3 by single crystal X-ray diffraction (SXRD) measurements. The discovery motivates the examination of three possible transition paths by comparing formation enthalpy with first-principles calculations and employing a custom- designed hydrogen detection setup. Furthermore, the most suitable adsorption position of O2 molecules on the (111) surfaces has been investigated by comparing the adsorption enthalpy from different candidate positions by utilizing first-principles calculations, implying the pivotal role of O2 molecules played in the rapid formation of La(OH)3 along the optimal transition path.
Article
16 January 2024Purification the Solvent for the Production of High-Density Polyethylene
The adsorption purification of gasoline fraction with NaX zeolites as a solvent for the production of high-density polyethylene at a large pilot plant with a layer height of the adsorbent layer from 1 to 8 m is considered. Removal of impurities of aromatic and unsaturated hydrocarbons, organosulfur impurities and water ensured the production of high-quality polyethylene. The main characteristics of the adsorption process (the dynamic activity of zeolite NaX, the length of the mass transfer zone) in a wide range of flow rates of the cleaned raw materials are determined, allowing the calculation of the adsorber without applying the principles of large-scale transition.
