Found 10 results
Article
03 April 2025Design, Building and Performance of a New Photocatalytic Reactor Using TiO2-Coated Rings Synthesized by Plasma Electrolytic Oxidation
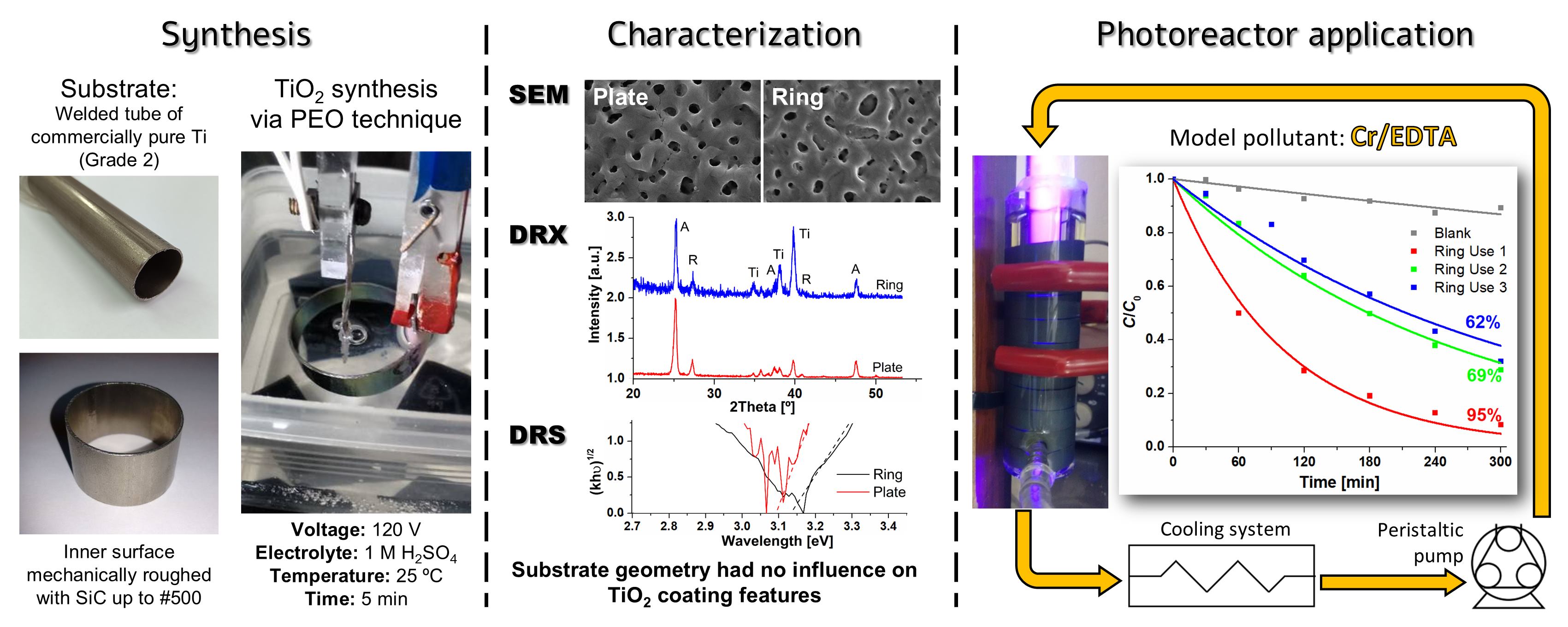
An annular UV photocatalytic reactor with recirculation in batch was designed and built. The design considered low construction, simple operation and maintenance costs, availability and durability of the materials used, easy cleaning, and high standards of hygiene and safety. The TiO2 photocatalysts were synthesized by plasma electrolytic oxidation (PEO) on commercial Ti rings were compared with coatings obtained on Ti plates as a reference, and no influence of the substrate geometry on the morphology, crystallinity, or bandgap of the coatings was observed. The efficiency of the photocatalytic reactor using 10 TiO2-coated rings was tested by Cr(VI) transformation in the presence of EDTA. The Cr(VI) transformation after 5 h irradiation attained 95%; a rather high photocatalytic activity (62%) was maintained after the third use of the rings without reactivation of the photocatalyst. These coatings synthesized by PEO have not been applied in modular photocatalytic reactors until now.
Perspective
01 April 2025Perspectives on the Development in the Selective Oxidation of Glycerol to Value-Added Chemicals via Photoelectrocatalysis Coupled with Hydrogen Evolution
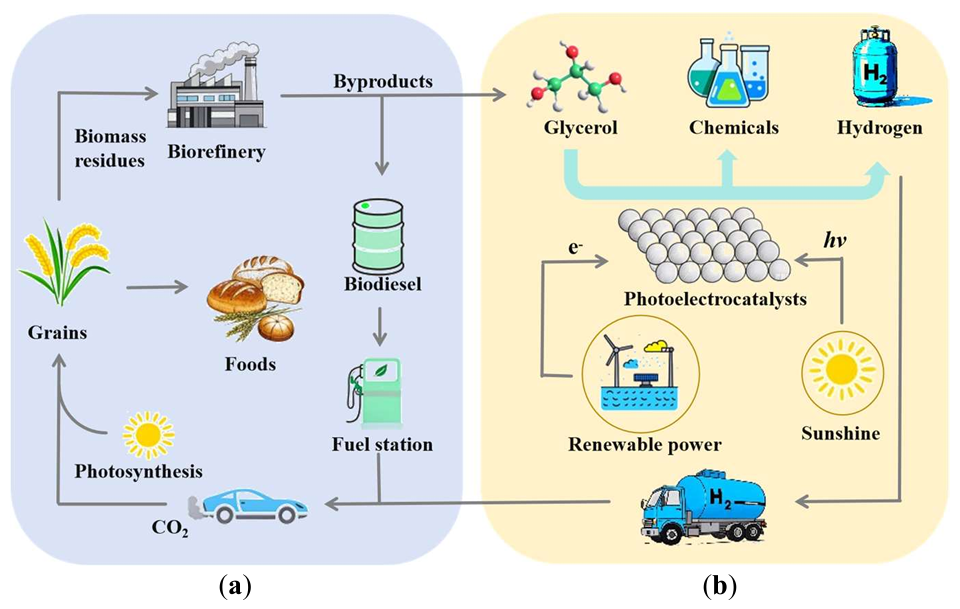
Harvesting sunlight to produce clean hydrogen fuel remains one of the main challenges for solving the energy crisis and ameliorating global warming. Photoelectrochemical (PEC) water splitting is considered to be a promising method for H2 production in the future. However, the efficiency still remains challenging due to the sluggish reaction dynamics for water oxidation. Recently, the thermodynamically favorable oxidation of glycerol in PEC systems has gained significant attention for its ability to produce value-added chemicals while simultaneously generating hydrogen. This process not only enhances the yield of high-value products but also minimizes energy consumption and reduces CO2 emissions. Valuable products from glycerol oxidation include 1,3-dihydroxyacetone (DHA), glyceraldehyde (GLD), tartronic acid (TA), formic acid (FA), and glyceric acid (GA). Thus, it is important to improve selectivity and productivity. In this work, we mainly summarize the recent research progress in improving the selectivity and productivity of glycerol upgrading products on the different photoanodes.
Research Highlight
31 March 2025Linear Semiconducting Polymers as Photoanodes for Oxidation Reactions
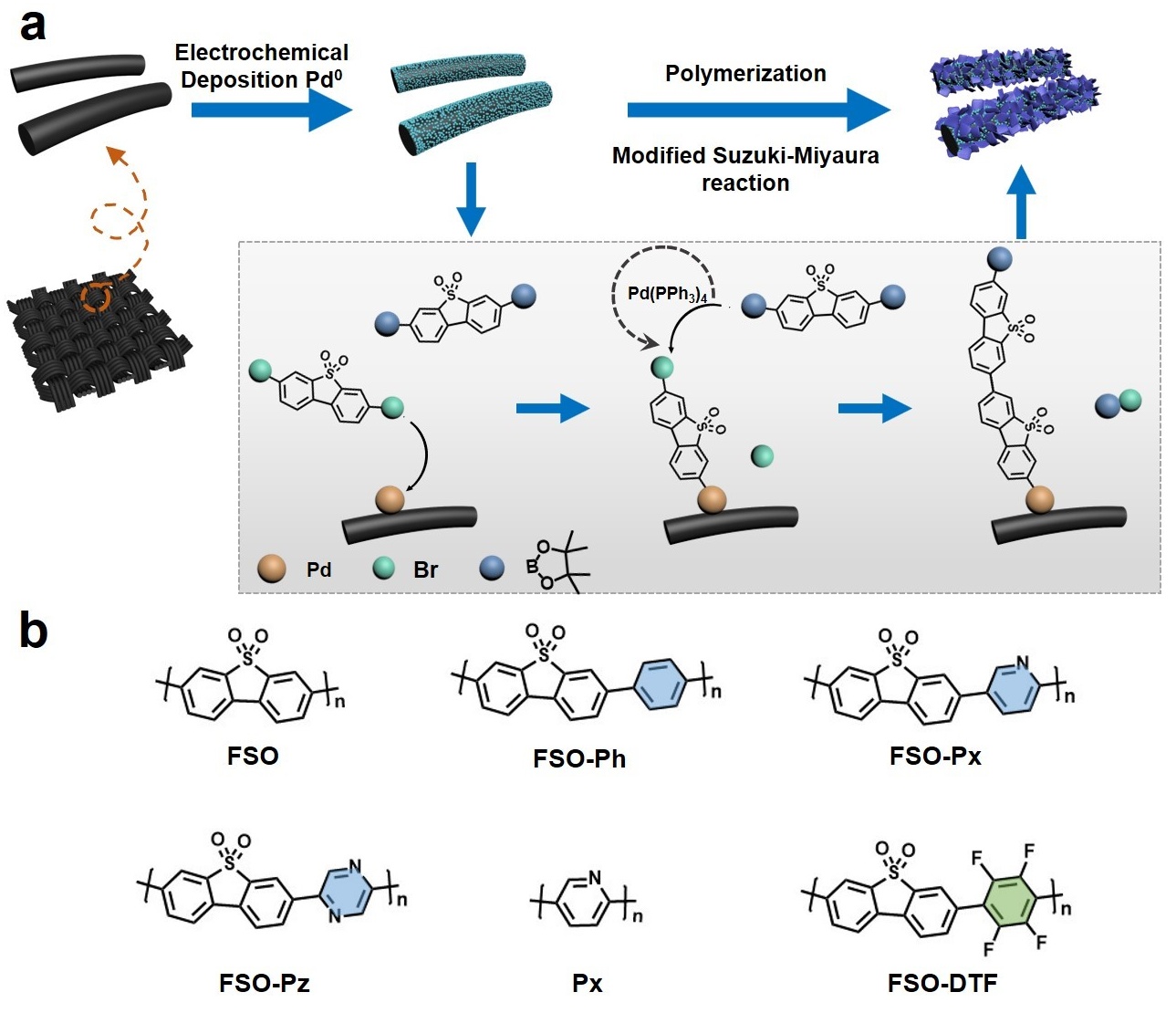
Photoelectrochemical (PEC) water splitting has attracted significant attention in the general field of photocatalysis. However, the high cost of constructing PEC systems limits their practical application. Recently, an innovative approach was proposed to synthesize linear semiconducting polymer-based films. The polymer structure was optimized for oxidation reactions. Furthermore, the active site of the optimal linear polymer was investigated through in-situ characterizations. This work has the potential to address the challenges of high material costs and polymer film development in PEC technology.
Review
06 March 2025A Comprehensive Review towards Oxidation Resistant Press-Hardened Steels
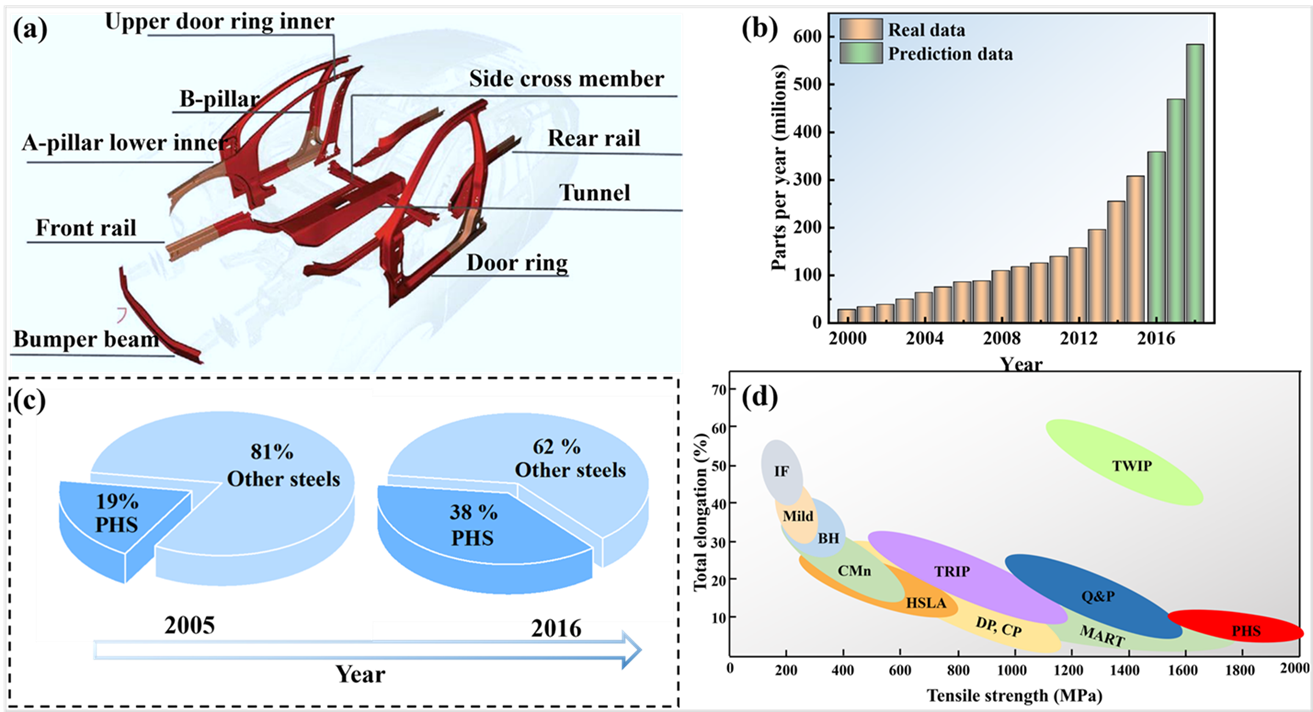
As an important lightweight material, press-hardened steels (PHS) are now widely used in the car body-in-white. However, severe oxidation of conventional Mn–B bare sheets not only damages production molds, but also prevents subsequent welding and painting, leading to a significant increase in production costs. The aim of this review is to systematically summarize the current solutions to overcome the problem of high-temperature oxidation of conventional Mn–B PHS and to highlight future directions for improvement. The review begins with a brief background on PHS, followed by a detailed description of measures to improve the oxidation resistance of conventional Mn–B PHS and the development of novel PHS with superior oxidation resistance. The oxidation resistance solutions for conventional Mn–B PHS mainly include the use of coatings and pre-deposited films. In contrast, the oxidation resistant PHS mainly includes the use of the oxidation resistant elements Cr, Si, Al or rare earth elements to improve the steel’s own high-temperature oxidation resistance.
Article
08 February 2025Degradation of Metformin Hydrochloride and Glibenclamide by Several Advanced Oxidation Processes
The degradation of metformin hydrochloride (MET) and glibenclamide (GLI), widely used anti-diabetics, was performed using an electrochemical advanced oxidation process, namely electro-Fenton, and several other Advanced Oxidation Processes (AOPs) of photocatalytic nature, like UV/H2O2, UV/persulfate, and UV/TiO2. The electrochemical behavior of the drugs was first characterized by cyclic and differential pulse voltammetry. The data implied that both drugs present quasi-reversible oxidation. The effect of the applied current and the airflow in the electrogeneration of hydrogen peroxide was studied. Degradations of 60% of the initial drug were obtained for aqueous solutions of 30 mg·L−1 of MET and 15 mg·L−1 of GLI by using photoelectron-Fenton conditions with 1.0 A of current and a Fe2+ concentration of 3.5 mg·L−1, although the removal of MET required 60 min of reaction while for GLI only 45 min were needed. The mineralization (organic carbon removal) percentages after 60 min of treatment were 20%and 30% for electro-Fenton and photo electro-Fenton processes, respectively. For UV/H2O2, UV/persulfate, and UV/TiO2 treatments of MET solutions, the order of observed degradations was UV/PS > UV/H2O2 > UV/TiO2 with maximum values of drug removal of 30% after 60 min of irradiation. This efficiency is lower than the removal observed with the electro-Fenton reaction. For GLI the order of degradation efficiency was UV/PS > UV/TiO2 > UV/H2O2, with maximum values of drug removals of 99.5% after only 10 min of irradiation. This performance is clearly better that in the case of electro-Fenton or photo-electro-Fenton. The removals of the two drugs when dissolved in chemical matrices that mimic real hospital wastewaters and seawater were also studied. They showed a clear dependency on the pharmaceutical of choice. While the degradation of MET was hampered by the presence of other chemicals in the two water matrices, GLI removal was remarkable, pointing towards a possible application in real wastewaters.
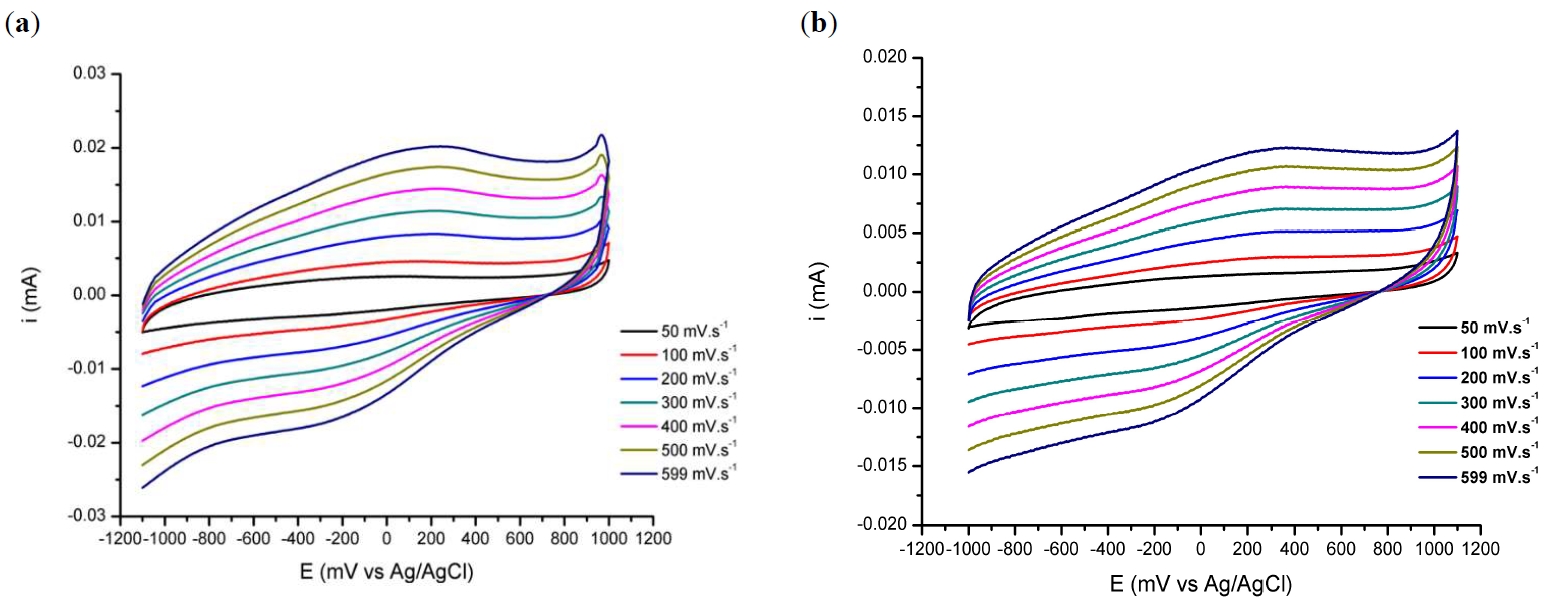
Article
21 January 2025Machining Characteristics of Graphene Oxide-Based Nanosuspensions in Abrasive Machining of Single-Crystal Si and SiC
Single-crystal silicon (Si) and silicon carbide (SiC) are core semiconductor materials in communication, lighting, power generation, and transportation. However, their high hardness and wear resistance combined with low fracture toughness have posed significant challenges for high-efficiency and low-damage machining. Aqueous suspensions containing nanoparticle additives have recently been developed for sustainable manufacturing due to their satisfactory tribological performance and environmentally friendly nature. In this work, nanoadditives, including two-dimensional (2D) graphene oxide (GO) nanosheets and zero-dimensional (0D) diamond nanoparticles, were ultrasonically dispersed in water to formulate different GO-based nanosuspensions for achieving high-efficiency and low-damage abrasive machining. The experimental results indicated that GO nanosuspension was a suitable coolant for grinding Si, generating a ground surface of 32 nm in Ra, owing to its great lubricity and excellent resistance against mechanical abrasion. Diamond-GO hybrid nanosuspension demonstrated a synergistic effect in abrasion, lubrication and oxidation, which was thus appropriate for polishing SiC single crystals, leading to approximate 60% and 30% improvements in removal and roughness respectively, in comparison to a commercially available diamond suspension.
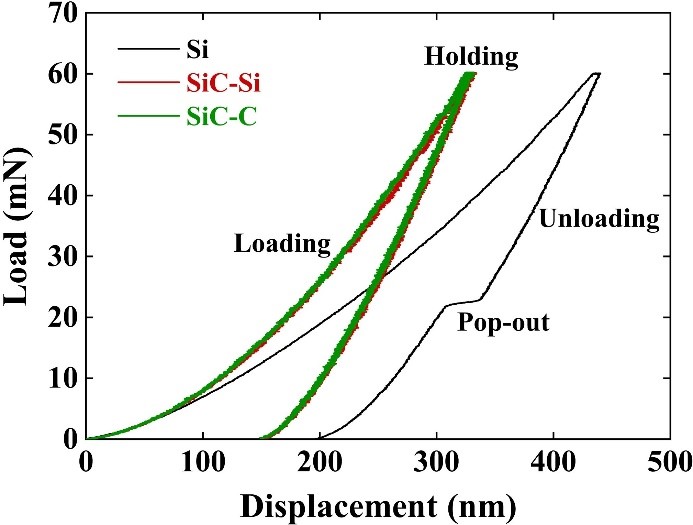
Review
30 December 2024
Mini Review on the Photocatalytic Removal of Gaseous Ammonia: Current Status
and Challenges
Ammonia
gas (NH3) is a notorious malodorous pollutant released mainly in
agriculture and industry. With the increasing demand for ammonia, environmental
pollution caused by ammonia discharge has seriously threatened human health and
safety. Due to the discrete emission and low concentration of NH3,
photocatalytic oxidation is an economical and efficient treatment strategy. TiO2,
as a common photocatalyst, has been widely used by researchers for the
photocatalytic removal of NH3. In addition, surface modification,
element doping, semiconductor recombination and metal loading are used to
improve the utilization rate of solar energy and carrier of TiO2 so
as to find a catalyst with high efficiency and high N2 selectivity.
Further, at present, there are three main removal mechanisms of NH3 photocatalytic oxidation: ·NH2 mechanism, iSCR mechanism and N2H4 mechanism. Among them, N2H4 mechanism is expected to be
the main removal path of NH3 photocatalytic oxidation in the future
because the removal process does not involve NOx and nitrate. This
review summarizes recent studies on the photocatalytic oxidation of NH₃,
focusing primarily on NH₃ removal efficiency, N₂ selectivity, and the underlying
removal mechanisms. Additionally, the potential future applications of NH₃
photocatalytic oxidation are discussed.
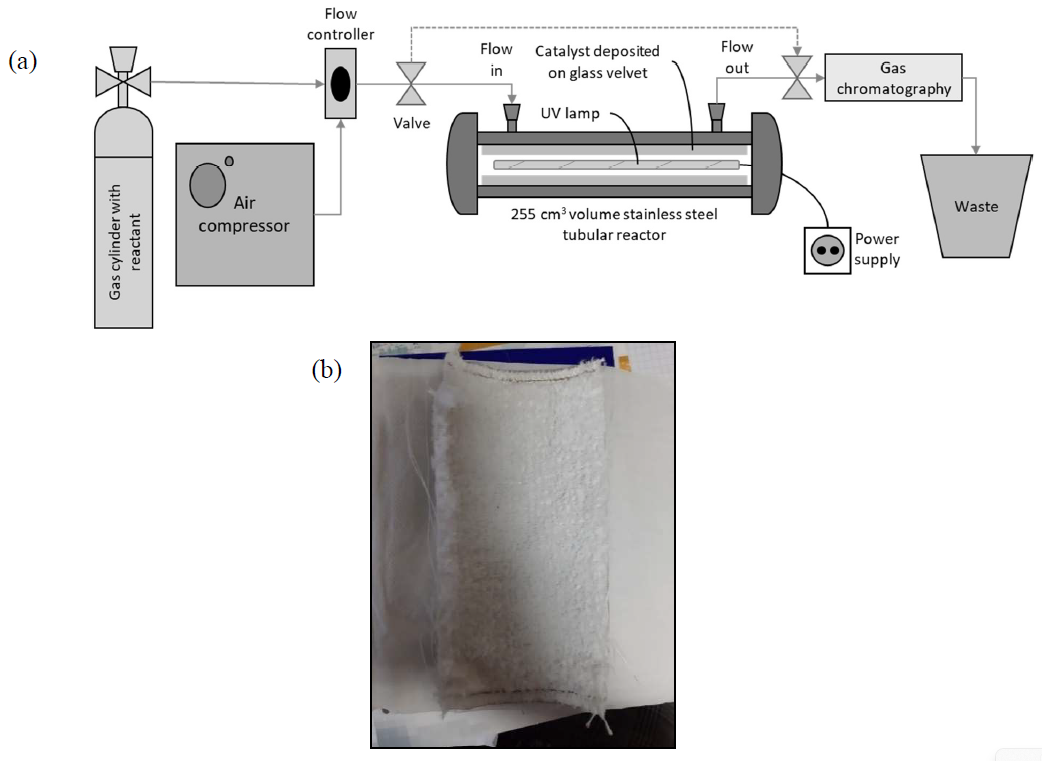
Article
28 August 2024Photocatalytic and Photothermal Catalytic Oxidation of Ethene and Ethanol Using TiO2-Based Catalysts under UV-C and UV-A Irradiation
Photocatalytic (PCO) and photothermocatalytic oxidation (PTCO) of ethene (C2H4) and ethanol (EtOH) are investigated using TiO2 and 1%Pt/TiO2 coating on velvet glass support in the presence of UV-A and UV-C irradiation. Both VOC are efficiently mineralised under UV-A irradiation and PCO, but the presence of Pt has a minor impact on their transformation. Instead, there is only a slight increase in the disappearance of EtOH and the formation of acetaldehyde, which are already observed in the dark. Surprisingly, when a higher photon flux is emitted with a UV-C lamp, photocatalytic disappearance and mineralisation of EtOH are less effective than under UV-A irradiation in the presence or absence of Pt. Similar behaviour is also observed on C2H4 PCO in the presence of 1%Pt/TiO2 but not on its PCO mineralisation with TiO2, which is improved by a factor equivalent to the number of photons emitted. Under PTCO, by increasing the temperature from 40 °C to 120 °C, only a benefit impact is observed on C2H4 and EtOH disappearance but an important decrease of mineralization of C2H4 was observed in presence of TiO2 and UV-C The behaviour of these two VOCs under different irradiations and temperatures will be discussed according to the catalytic process.
Review
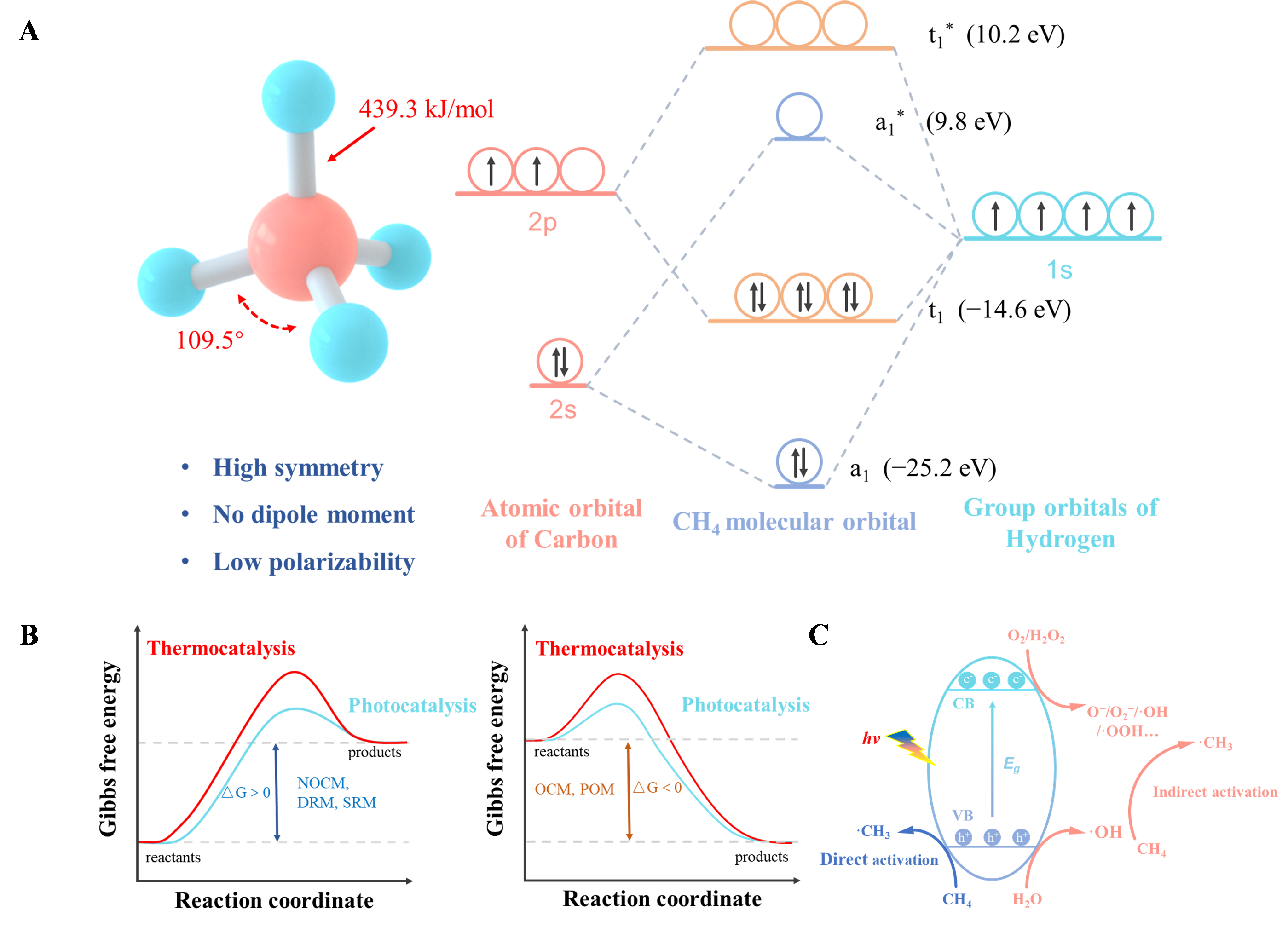
11 July 2024Photocatalytic Aerobic Conversion of Methane
The direct conversion of methane into high-value chemicals has been a persistent research focus in the fields of chemical engineering and energy. Photocatalysis, as an innovative technology, not only circumvents the issues of catalyst sintering and carbon deposition associated with traditional thermal catalysis but also transcends thermodynamic limitations by providing new reaction pathways. Utilizing molecular oxygen as an oxidant generates various reactive oxygen species, offering unique thermodynamic advantages for methane conversion. This review summarizes the advancements in photocatalytic partial oxidation (PPOM) and oxidative coupling of methane (POCM) using oxygen as an oxidant. It discusses the activation mechanisms and reaction pathways of methane and oxygen in different systems, as well as the application of photochemical cycling strategies in methane conversion. Finally, it addresses the challenges in this field, proposes potential solutions, and offers perspectives on the future development of photocatalytic systems.
Article
17 April 2024Thermogravimetric Study of the Oxidation Behavior of the Cantor’s Alloy at 1000 °C and Beyond
A polycrystalline Cantor alloy, equimolar in Co, Cr, Fe, Mn and Ni, was cast. It was subjected to oxidation in a thermo-balance in a flow of synthetic dry air, at 1000, 1050, 1100 and 1150 °C. The mass gain was globally parabolic but rather irregular. The parabolic constants, ranging from 55 to 700 × 10−12·g2·cm−4·s−1, are much higher than for a chromia-forming alloy. They obey an Arrhenius law with an activation energy equal to 270 kJ/mol. The external oxide scales formed are composed of an outer part made of manganese oxide and an inner part made of (Cr, Mn) oxide containing a thin internal layer of chromia. The Mn and Cr-depleted depths and the Mn and Cr masses lost by the alloy increase with the oxidation temperature. Cr-rich acicular particles precipitated in subsurface at 1100 °C and internal oxidation along the grain boundaries are present in the whole thickness of the sample oxidized at 1150 °C. Oxide spallation occurred during the cooling, at temperatures in the 200–350 °C range, only for the alloys oxidized at 1050 and 1100 °C. Not too thick scale (1000 °C) or deep internal oxidation (1150 °C) may be favorable for scale adherence.
