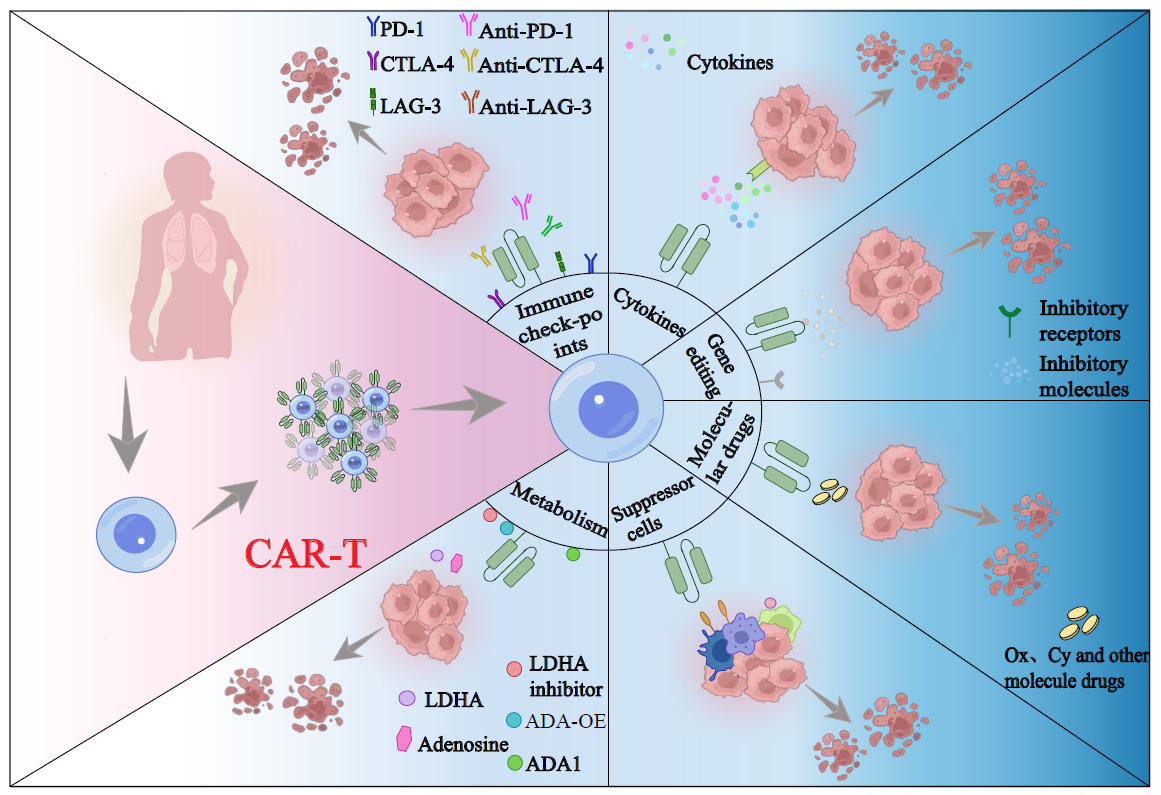
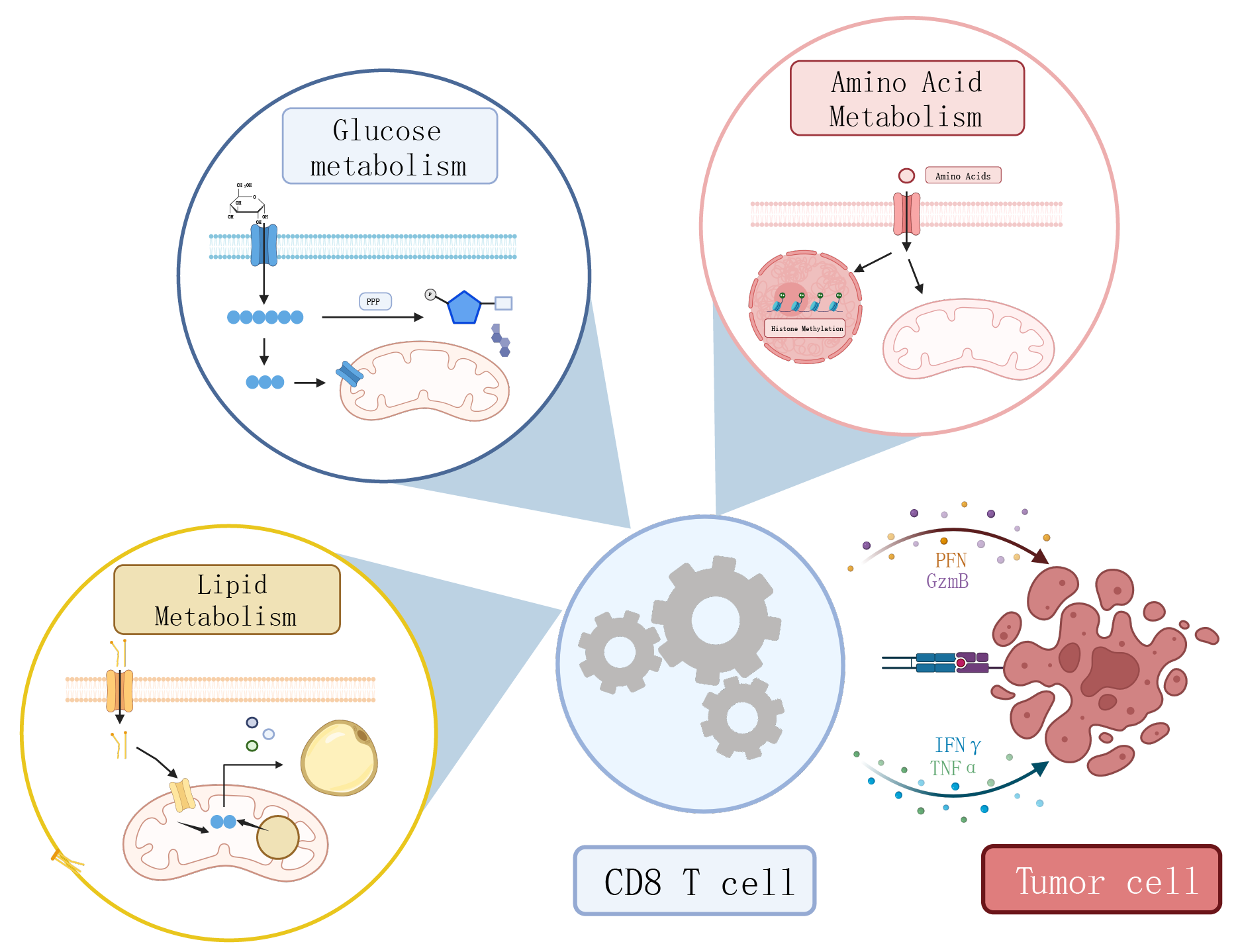
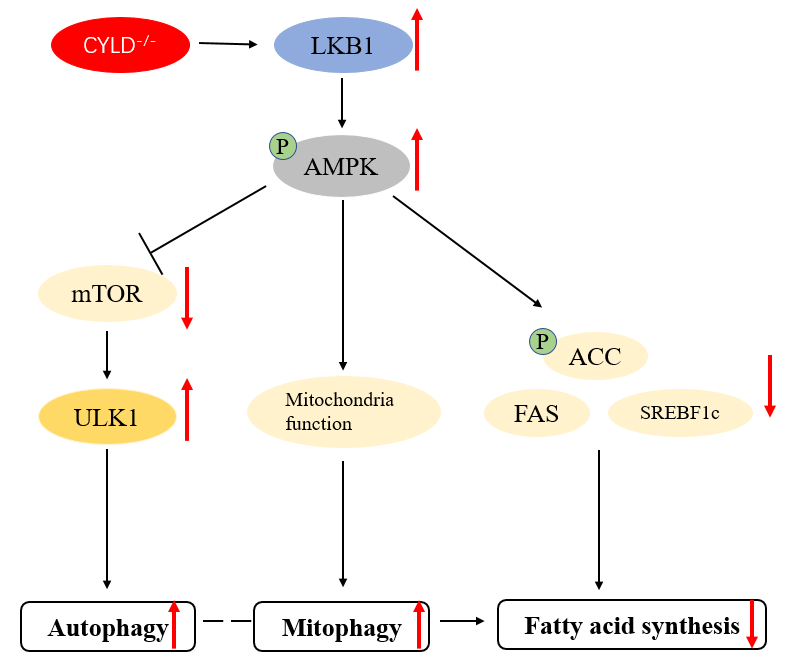
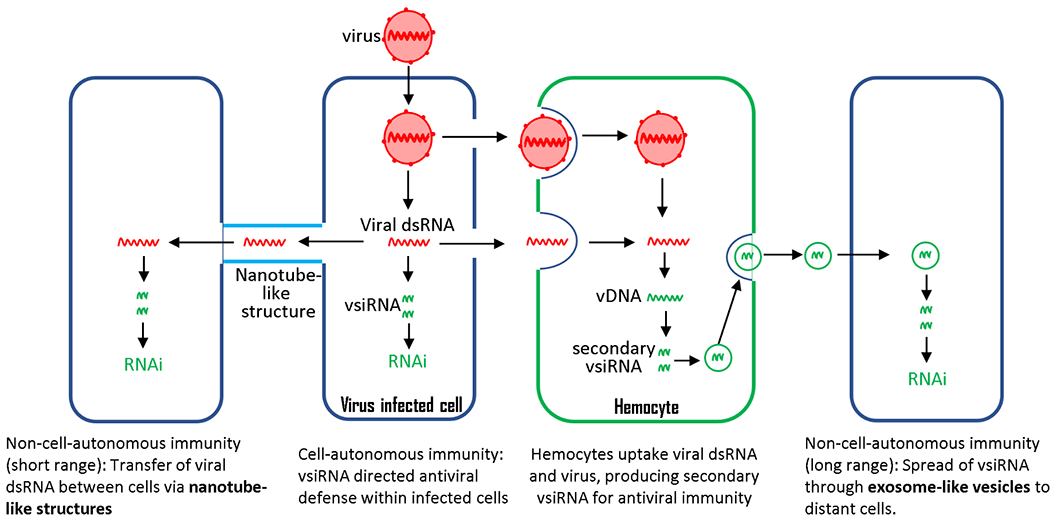
Cytotoxic CD8 T cells play a crucial role in controlling
tumor progression. However, T cells infiltrating tumor tissues upregulate
inhibitory receptors, reduce cytokine secretion, and lose their killing
function, a state known as exhaustion. Thus, preventing or reversing T cell
exhaustion is essential for sustaining a successful antitumor immune response.
Recent studies have shown that T cell immunity not only requires the three
primary signals—antigen
receptor signaling, costimulation, and cytokines—but
is largely shaped by endogenous and ambient metabolites as a fourth regulatory
signal. Therefore, metabolic changes in the tumor microenvironment, caused by
tumor cell proliferation and tissue remodeling, have a significant impact on
the function of tumor-infiltrating T cells. This paper will review mechanisms
by which three major types of metabolites—carbohydrates, lipids, and amino acids—influence T cell
exhaustion in the tumor microenvironment, providing insights and directions for
exploring metabolic targets in antitumor immunity.utf-8
 Open Access
Open Access